UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-Q
| QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the Quarterly Period Ended June 30, 2025
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the Transition Period from __________________ to _________________________
Commission File Number 001-33650
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | ||||
| (Address of principal executive offices) | (zip code) | ||||
Registrant’s telephone number, including area code: 908 -842-0100
| Securities registered pursuant to Section 12(b) of the Act: | ||||||||
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||
The | ||||||||
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§ 232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See definition of “large accelerated filer,” “accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act:
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |||||||||||
| ☒ | Smaller reporting company | |||||||||||||
| Emerging growth company | ||||||||||||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes ☐ No ☒
Indicate the number of shares outstanding of each of the issuer's classes of common stock, as of the latest practicable date.
| Class | Outstanding as of August 7, 2025 | |||||||
| Common stock, $0.001 par value per share | shares | |||||||
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This quarterly report (this “Quarterly Report”) contains “forward-looking” statements within the meaning of the Private Securities Litigation Reform Act of 1995, as well as historical information. When used in this Quarterly Report, statements that are not statements of current or historical fact may be deemed to be forward-looking statements, including, without limitation, all statements related to any expectations of revenues, expenses, cash flows, earnings or losses from operations, cash required to maintain current and planned operations, capital or other financial items; any statements of the plans, strategies and objectives of management for future operations; any plans or expectations with respect to product research, development and commercialization, including regulatory approvals; any other statements of expectations, plans, intentions or beliefs; and any statements of assumptions underlying any of the foregoing. Without limiting the foregoing, the words “plan,” “project,” “forecast,” “outlook,” “intend,” “may,” “will,” “expect,” “anticipate,” “likely,” “believe,” “could,” “anticipate,” “estimate,” “continue,” “target” or similar expressions or other variations or comparable terminology are intended to identify such forward-looking statements, although some forward-looking statements are expressed differently. We remind readers that forward-looking statements are merely predictions and therefore inherently subject to uncertainties and other factors and involve known and unknown risks that could cause the actual results, performance, levels of activity or our achievements or industry results, to be materially different from any future results, performance, levels of activity or our achievements or industry results expressed or implied by such forward-looking statements. Factors that could cause our actual results to differ materially from anticipated results expressed or implied by forward-looking statements include, among others:
•our ability to obtain sufficient capital or strategic business arrangements to fund our operations and expansion plans, including collecting amounts owed to us under various licensing and other strategic arrangements, meeting our financial obligations under various licensing and other strategic arrangements, the funding of our clinical trials for product candidates, and the commercialization of the relevant technology;
•our ability to build and maintain the management and human resources infrastructure necessary to support the operation and/or growth of our business;
•whether a market is established for our products and our ability to capture a meaningful share of this market;
•scientific, regulatory and medical developments beyond our control;
•our ability to obtain and maintain, as applicable, appropriate governmental licenses, accreditations or certifications or to comply with healthcare laws and regulations or any other adverse effect or limitations caused by government regulation of our business;
•whether any of our current or future patent applications result in issued patents, the scope of those patents and our ability to obtain and maintain other rights to technology required or desirable for the conduct of our business, and our ability to commercialize products without infringing upon the claims of third-party patents;
•whether any potential strategic or financial benefits of various licensing agreements will be realized;
•our ability to diversify our pipeline of development product candidates, which could include an acquisition, merger, business combination, in-license or other strategic transaction, and whether any of such efforts will result in us entering into or completing any transaction or that any such transaction, if completed, will add to shareholder value;
•the results of our development activities;
•our ability to complete our other planned clinical trials (or initiate other trials) in accordance with our estimated timelines due to delays associated with enrolling patients due to the novelty of the treatment, the size of the patient population, competition with other clinical trials for similar subjects, patient and/or investigator site availability and accessibility due to external macroenvironmental factors and the need of patients to meet the inclusion criteria of the trial or otherwise;
•the extent to which any future public health crisis and their long-term effects may impact, directly or indirectly, our business, including our clinical trials and financial condition; and
•other factors discussed in “Risk Factors” in our Annual Report on Form 10-K filed with the Securities and Exchange Commission (the “SEC”) on February 27, 2025 (our “2024 Form 10-K”).
The factors discussed herein, including those risks described in “Item 1A. Risk Factors” and elsewhere in our 2024 Form 10-K and in our other periodic filings with the SEC, which are available for review at www.sec.gov, could cause actual results and developments to be materially different from those expressed or implied by such statements. All forward-looking statements attributable to us are expressly qualified in their entirety by these and other factors. Readers are cautioned not to place undue
2
reliance on these forward-looking statements, which speak only as of the date they were made. Except as required by law, we undertake no obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise.
3
TABLE OF CONTENTS
PART I- FINANCIAL INFORMATION | Page No. | |||||||
Item 1. | ||||||||
Notes to Unaudited Consolidated Financial Statements | ||||||||
Item 2. | Management’s Discussion and Analysis of Financial Condition and Results of Operations | |||||||
Item 3. | Quantitative and Qualitative Disclosures About Market Risk | |||||||
Item 4. | Controls and Procedures | |||||||
PART II- OTHER INFORMATION | ||||||||
Item 1. | Legal Proceedings | |||||||
Item 1A. | Risk Factors | |||||||
Item 2. | Unregistered Sales of Equity Securities and Use of Proceeds | |||||||
Item 3. | Defaults Upon Senior Securities | |||||||
Item 4. | Mine Safety Disclosures | |||||||
Item 5. | Other Information | |||||||
Item 6. | Exhibits | |||||||
Signatures | ||||||||
4
PART I. FINANCIAL INFORMATION
ITEM 1. FINANCIAL STATEMENTS
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
(In thousands, except share data)
| June 30, 2025 | December 31, 2024 | ||||||||||
| ASSETS | (Unaudited) | ||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
Marketable securities | |||||||||||
| Accounts receivable | |||||||||||
| Prepaid and other current assets | |||||||||||
| Total current assets | |||||||||||
| Property and equipment, net | |||||||||||
| Acquired license - intangible, net | |||||||||||
| Other assets | |||||||||||
| Total assets | $ | $ | |||||||||
| LIABILITIES, NON-CONTROLLING INTERESTS AND STOCKHOLDERS' EQUITY | |||||||||||
| Liabilities | |||||||||||
| Accounts payable | $ | $ | |||||||||
| Accrued liabilities | |||||||||||
| Total current liabilities | |||||||||||
| Other long-term liabilities | |||||||||||
| Total liabilities | |||||||||||
| Commitments and Contingencies (Note 14) | |||||||||||
| Stockholders' Equity | |||||||||||
Common stock, $ and outstanding, respectively | |||||||||||
| Additional paid-in capital | |||||||||||
Treasury stock, at cost; | ( | ( | |||||||||
| Accumulated deficit | ( | ( | |||||||||
| Accumulated other comprehensive loss | ( | ( | |||||||||
| Total Lisata Therapeutics, Inc. stockholders' equity | |||||||||||
| Non-controlling interests | ( | ( | |||||||||
| Total equity | |||||||||||
| Total liabilities, non-controlling interests and stockholders' equity | $ | $ | |||||||||
See accompanying notes to consolidated financial statements.
5
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF OPERATIONS
(Unaudited)
(In thousands, except per share data)
| Three Months Ended June 30, | Six Months Ended June 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| Revenue | $ | $ | $ | $ | |||||||||||||||||||
| Operating Expenses: | |||||||||||||||||||||||
| Research and development | |||||||||||||||||||||||
| General and administrative | |||||||||||||||||||||||
| Total operating expenses | |||||||||||||||||||||||
| Operating loss | ( | ( | ( | ( | |||||||||||||||||||
| Other income (expense): | |||||||||||||||||||||||
| Investment income, net | |||||||||||||||||||||||
| Other expense, net | ( | ( | ( | ( | |||||||||||||||||||
| Total other income | |||||||||||||||||||||||
| Net loss before benefit from income taxes and noncontrolling interests | ( | ( | ( | ( | |||||||||||||||||||
| Benefit from income taxes | ( | ( | |||||||||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
| Less - net income (loss) attributable to noncontrolling interests | |||||||||||||||||||||||
| Net loss attributable to Lisata Therapeutics, Inc. common stockholders | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
| Basic and diluted loss per share | |||||||||||||||||||||||
| Lisata Therapeutics, Inc. common stockholders | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
Weighted average common shares outstanding | |||||||||||||||||||||||
Basic and diluted shares | |||||||||||||||||||||||
See accompanying notes to consolidated financial statements.
6
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(Unaudited)
(In thousands)
| Three Months Ended June 30, | Six Months Ended June 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
| Other comprehensive gain (loss): | |||||||||||||||||||||||
| Available for sale securities - net unrealized gain (loss) | ( | ||||||||||||||||||||||
| Cumulative translation adjustment arising during the period | ( | ||||||||||||||||||||||
| Total other comprehensive gain (loss) | ( | ||||||||||||||||||||||
| Comprehensive loss attributable to Lisata Therapeutics, Inc. common stockholders | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
See accompanying notes to consolidated financial statements.
7
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF EQUITY
(Unaudited)
(In thousands)
| Common Stock | Additional Paid-in Capital | Accumulated Other Comprehensive Loss | Accumulated Deficit | Treasury Stock | Total Lisata Therapeutics, Inc. Stockholders' Equity | Non- Controlling Interest in Subsidiary | Total Equity | |||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at March 31, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | ( | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Net proceeds from issuances of common stock | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized gain on marketable securities | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at June 30, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Common Stock | Additional Paid-in Capital | Accumulated Other Comprehensive Loss | Accumulated Deficit | Treasury Stock | Total Lisata Therapeutics, Inc. Stockholders' Equity | Non- Controlling Interest in Subsidiary | Total Equity | |||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2023 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | ( | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Net proceeds from issuances of common stock | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized loss on marketable securities | — | — | — | ( | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | — | ( | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Balance at June 30, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
8
| Common Stock | Additional Paid-in Capital | Accumulated Other Comprehensive Loss | Accumulated Deficit | Treasury Stock | Total Lisata Therapeutics, Inc. Stockholders' Equity | Non- Controlling Interest in Subsidiary | Total Equity | |||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at March 31, 2025 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | ( | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | ( | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Net proceeds from issuances of common stock | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized gain on marketable securities | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at June 30, 2025 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Common Stock | Additional Paid-in Capital | Accumulated Other Comprehensive Loss | Accumulated Deficit | Treasury Stock | Total Lisata Therapeutics, Inc. Stockholders' Equity | Non- Controlling Interest in Subsidiary | Total Equity | |||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | ( | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Net proceeds from issuances of common stock | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Proceeds from option exercise | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized gain on marketable securities | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Foreign currency translation adjustment | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at June 30, 2025 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||
9
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
(Unaudited)
(In thousands)
| Six Months Ended June 30, | |||||||||||
| 2025 | 2024 | ||||||||||
| Cash flows from operating activities: | |||||||||||
| Net loss | $ | ( | $ | ( | |||||||
| Adjustments to reconcile net loss to net cash used in operating activities: | |||||||||||
| Share-based compensation | |||||||||||
| Depreciation and amortization | |||||||||||
| Loss from equity method investment | |||||||||||
| Amortization/accretion on marketable securities | ( | ( | |||||||||
| Changes in operating assets and liabilities: | |||||||||||
| Accounts receivable | |||||||||||
| Prepaid and other current assets | ( | ||||||||||
| Other assets | |||||||||||
| Accounts payable, accrued liabilities and other liabilities | ( | ( | |||||||||
| Net cash used in operating activities | ( | ( | |||||||||
| Cash flows from investing activities: | |||||||||||
| Purchase of marketable securities | ( | ( | |||||||||
| Sale of marketable securities | |||||||||||
| Purchase of property and equipment | ( | ||||||||||
| Investment in Impilo Therapeutics | ( | ||||||||||
| Net cash provided by investing activities | |||||||||||
| Cash flows from financing activities: | |||||||||||
| Proceeds from exercise of options | |||||||||||
| Tax withholding payments on net share settlement equity awards | ( | ( | |||||||||
| Net proceeds from issuance of common stock | |||||||||||
| Net cash provided by (used in) financing activities | ( | ||||||||||
| Effect of exchange rate changes on cash | ( | ||||||||||
| Net increase in cash and cash equivalents | |||||||||||
| Cash and cash equivalents at beginning of period | |||||||||||
| Cash and cash equivalents at end of period | $ | $ | |||||||||
See accompanying notes to consolidated financial statements.
10
LISATA THERAPEUTICS, INC. AND SUBSIDIARIES
NOTES TO UNAUDITED CONSOLIDATED FINANCIAL STATEMENTS
Note 1 – Description of Business
Overview
Lisata Therapeutics, Inc. (together with its subsidiaries, the “Company”) is a clinical-stage pharmaceutical company dedicated to the discovery, development, and commercialization of innovative therapies for the treatment of solid tumors and other major diseases. The Company's investigational product, certepetide (formerly known as LSTA1 or CEND-1), is designed to activate a novel uptake pathway that allows co-administered or tethered (i.e., molecularly bound) anti-cancer drugs to target and penetrate solid tumors more effectively. Certepetide actuates this active transport system in a tumor-specific manner, resulting in systemically co-administered anti-cancer drugs more efficiently penetrating and accumulating in the tumor, while normal tissues are expected to remain unaffected. Certepetide has also been shown to modify the tumor microenvironment (“TME”) by reducing T-regulatory cells and augmenting cytotoxic T cells, thereby making tumors more susceptible to immunotherapies while also inhibiting the metastatic cascade (i.e., the spread of cancer to other parts of the body). The Company, its collaborators and other researchers have amassed and continue to amass significant non-clinical data demonstrating enhanced delivery of a range of existing and emerging anti-cancer therapies, including chemotherapeutics, immunotherapies, and RNA-based therapeutics. To date, certepetide has also demonstrated favorable safety, tolerability and activity in completed and ongoing clinical trials designed to enhance delivery of standard-of-care chemotherapy for pancreatic cancer. The Company is exploring certepetide as a means to enable a variety of therapeutic modalities to treat a range of solid tumors more effectively. Currently, certepetide is the subject of several Phase 2 clinical studies being conducted globally in a variety of solid tumor types, including metastatic pancreatic ductal adenocarcinoma (mPDAC), cholangiocarcinoma, appendiceal cancer, colon cancer and glioblastoma multiforme in combination with a variety of anti-cancer regimens.
The Company's leadership team has amassed several decades of collective biopharmaceutical and pharmaceutical product development experience across a variety of therapeutic categories and at all stages of development, ranging from preclinical through to product registration and launch. The Company's goal is to develop and commercialize products that address important unmet medical needs.
The Company has a history of net operating losses and negative cash flows from its operating activities, and has cash, cash equivalents and marketable securities of approximately $22.0 million as of June 30, 2025. The Company anticipates incurring additional losses until such time, if ever, that it can generate significant revenues from its products currently in development. To manage capital for operating needs in the short-term, as previously disclosed, initiation of the FORTIFIDE study remains on hold and the Company has delayed commencement of certain Phase 3 readiness activities as it relates to chemistry, manufacturing and controls (“CMC”). The Company’s ongoing trials continue to progress as planned. The Company also identified certain operating efficiencies which had a positive impact on the cash flow forecast. The Company believes that, as a result, it currently has sufficient cash to meet its funding requirements over the next year. To meet our long-term liquidity needs, the Company expects that it will need additional financing by the fourth quarter of 2026, which could result in additional potential issuances of debt or equity securities in public or private financings, partnerships and/or collaborations and/or sale of assets.
Basis of Presentation
The accompanying unaudited Consolidated Financial Statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”) for interim financial information and with the instructions to Form 10-Q and Article 10 of Regulation S-X of the SEC for interim financial information. Accordingly, they do not include all of the information and footnotes required by generally accepted accounting principles for complete financial statements. In the opinion of management, the accompanying Consolidated Financial Statements of the Company and its subsidiaries, which are unaudited, include all normal and recurring adjustments considered necessary to present fairly the Company’s financial position as of June 30, 2025, and the results of its operations and its cash flows for the periods presented. The unaudited consolidated financial statements herein should be read together with the historical consolidated financial statements of the Company for the years ended December 31, 2024 and 2023 included in our 2024 Form 10-K. Operating results for the three and six months ended June 30, 2025 are not necessarily indicative of the results that may be expected for the year ending December 31, 2025.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the
11
date of the consolidated financial statements. Estimates also affect the reported amounts of expenses during the reporting period. The Company bases its estimates on historical experience and other assumptions believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. The Company makes critical estimates and assumptions in determining stock-based awards values. Accordingly, actual results could differ from those estimates and assumptions.
Segment Information
The Company operates as one operating segment, the research and development of its investigational drug product. The Company's Chief Operating Decision Maker (“CODM”) is its Chief Executive Officer, who manages the business on a consolidated basis.
Principles of Consolidation
Foreign Currency Remeasurement
The Company’s reporting currency is the U.S. Dollar. The functional currency of Lisata Therapeutics Australia Pty Ltd., which is a foreign subsidiary of the Company, is the Australian Dollar. The assets and liabilities of Lisata Therapeutics Australia Pty Ltd. are translated into U.S. Dollars at the exchange rates in effect at each balance sheet date, and the results of operations are translated using the average exchange rates prevailing throughout the reporting period. Adjustments resulting from translating foreign functional currency financial statements into U.S. Dollars are included in the foreign currency translation adjustment, a component of accumulated other comprehensive income (loss) in stockholders' equity.
Note 2 – Summary of Significant Accounting Policies
Cash and Cash Equivalents
Concentration of Risks
The Company is subject to credit risk from its portfolio of cash, cash equivalents, accounts receivable and marketable securities. Under its investment policy, the Company limits amounts invested in such securities by credit rating, maturity, industry group, investment type and issuer, except for securities issued by the U.S. government, thereby reducing credit risk exposure. Cash is held at major banks in the United States and may exceed federally insured limits. The Company does not believe that it is subject to unusual credit risk beyond the normal credit risk associated with commercial banking relationships. The goals of the Company's investment policy, in order of priority, are as follows: safety and preservation of principal and diversification of risk, liquidity of investments sufficient to meet cash flow requirements, and a competitive after-tax rate of return. The Company’s accounts receivable balance as of June 30, 2025 and December 31, 2024 is primarily derived from its license agreement dated November 30, 2024, more fully described in Note 17. The Company has no significant off-balance sheet concentrations of credit risk, such as foreign currency exchange contracts, option contracts, or other hedging arrangements.
Accounts Receivable
12
Marketable Securities
The Company determines the appropriate classification of its marketable securities at the time of purchase and reevaluates such designation at each balance sheet date. All of the Company's marketable securities are considered as available-for-sale and carried at estimated fair values and reported in cash equivalents and marketable securities. Unrealized gains and losses on available-for-sale securities, that are not the result of credit losses, are excluded from net income and reported in accumulated other comprehensive income (loss) as a separate component of stockholders' equity. Other income (expense), net, includes interest, dividends, amortization of purchase premiums and discounts, realized gains and losses on sales of securities and credit losses recognized through an allowance for credit losses, if any. The cost of securities sold is based on the specific identification method. The Company regularly reviews all of its investments for other-than-temporary declines in fair value. The Company's review includes the consideration of the cause of the impairment, including the creditworthiness of the security issuers, the number of securities in an unrealized loss position, the severity and duration of the unrealized losses, whether the Company has the intent to sell the securities and whether it is more likely than not that it will be required to sell the securities before the recovery of their amortized cost basis. When the Company determines that the decline in fair value of an investment is below its accounting basis and this decline is other-than-temporary, it reduces the carrying value of the security it holds and records a loss for the amount of such decline.
Property and Equipment
| Furniture and fixtures | |||||
| Computer equipment | |||||
| Software | |||||
| Leasehold improvements | Shorter of useful life or lease term | ||||
Long-lived Assets
Long-lived assets consist of property and equipment. The assets are amortized on a straight-line basis over their respective useful lives. The Company reviews long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset exceeds the fair value of the asset. If other events or changes in circumstances indicate that the carrying amount of an asset that the Company expects to hold and use may not be recoverable, the Company will estimate the undiscounted future cash flows expected to result from the use of the asset and/or its eventual disposition, and recognize an impairment loss, if any. The impairment loss, if determined to be necessary, would be measured as the amount by which the carrying amount of the assets exceeds the fair value of the assets.
Share-Based Compensation
Loss Per Share
13
Treasury Stock
Treasury stock purchases are accounted for under the cost method whereby the entire cost of the acquired stock is recorded as treasury stock. Gains or losses on the subsequent reissuance of shares are credited or charged to additional paid in capital.
Research and Development Costs
Research and development (“R&D”) expenses include salaries, benefits, and other headcount related costs, clinical trial and related clinical manufacturing costs, contract and other outside service fees including sponsored research agreements, and facilities and overhead costs. The Company expenses the costs associated with research and development activities when incurred.
To further drive the Company’s initiatives, the Company will continue targeting key governmental agencies and not-for-profit organizations to contribute funds for the Company’s research and development programs. The Company accounts for such grants as a deduction to the related expense in research and development operating expenses when earned.
In-process Research and Development Expense
Upfront payments that relate to the acquisition of a new drug compound, as well as pre-commercial milestone payments, are immediately expensed as IPR&D in the period in which they are incurred, provided that the new drug compound did not also include processes or activities that would constitute a “business” as defined under U.S. GAAP, the drug has not achieved regulatory approval for marketing and, absent obtaining such approval, has no established alternative future use. The Company accounts for contingent consideration payable upon achievement of certain regulatory, development or sales milestones in such asset acquisitions when the underlying contingency is probable and estimable. Milestone payments made to third parties subsequent to regulatory approval will be capitalized as intangible assets and amortized over the estimated remaining useful life of the related product.
Intangible Asset
The Company’s intangible asset consists of a single asset, a license agreement with Qilu Pharmaceutical, Co., Ltd. (“Qilu”) acquired in the Company's acquisition of Cend Therapeutics, Inc (the “Cend Merger”), with a value of $0.4 million. The intangible asset is stated at fair value and is amortized using the straight-line method over its estimated useful life of 5.00 years. Amortization expense was $18 thousand and $35 thousand for the three and six months ended June 30, 2025, respectively, and $18 thousand and $35 thousand for the three and six months ended June 30, 2024, respectively. The intangible asset is reviewed for potential impairment when events or circumstances indicate that carrying amounts may not be recoverable. The projected amortization expense is $71
Revenue Recognition
The Company evaluates license and collaboration arrangements to determine whether units of account within the arrangement exhibit the characteristics of a vendor and customer relationship. For arrangements and units of account where a customer relationship exists, the Company applies the revenue recognition guidance. The Company recognizes revenue upon the transfer of promised goods or services to customers in an amount that reflects the consideration to which the Company expects to be entitled in exchange for those goods or services. To determine revenue recognition for contracts with customers the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the Company satisfies the performance obligations. At contract inception, the Company assesses the goods or services promised within each contract and assesses whether each promised good or service is distinct and determines those that are performance obligations. The Company then recognizes as revenue the amount of the transaction price that is allocated to the respective performance obligation when (or as) the performance obligation is satisfied. Taxes imposed by governmental authorities on the Company's revenue, such as sales taxes and withholding taxes, are excluded from net revenue.
14
Milestones
At the inception of each arrangement that includes milestone payments (variable consideration), the Company evaluates whether the milestones are considered probable of being reached and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable that a significant revenue reversal would not occur, the associated milestone value is included in the transaction price. Milestone payments that are not within the Company or the Company’s collaboration partner’s control, such as regulatory approvals, are generally not considered probable of being achieved until those approvals are received. The transaction price is then allocated to each performance obligation on a relative stand-alone selling price basis, for which the Company recognizes revenue as or when the performance obligations under the contract are satisfied. At the end of each subsequent reporting period, the Company re-evaluates the probability of achievement of such milestones and any related constraint, and if necessary, adjusts the Company’s estimate of the overall transaction price. Any such adjustments are allocated on a cumulative catch-up basis to satisfied and partially satisfied performance obligations, with the consideration allocated to an ongoing performance obligation being recognized over the period of performance. For the three and six months ended June 30, 2025 and June 30, 2024, the Company has not
Royalties
For arrangements that include sales-based royalties, including milestone payments based on the level of sales, and for which the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (i) when the related sales occur, or (ii) when the performance obligation to which some or all of the royalty has been allocated has been satisfied (or partially satisfied). To date, the Company has not recognized any royalty revenue from any collaborative arrangement.
Note 3 – Available-for-Sale Securities
The following table is a summary of available-for-sale securities recorded in cash and cash equivalents or marketable securities in the Company's Consolidated Balance Sheets (in thousands):
| June 30, 2025 | December 31, 2024 | ||||||||||||||||||||||||||||||||||||||||||||||
| Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | Cost | Gross Unrealized Gains | Gross Unrealized Losses | Estimated Fair Value | ||||||||||||||||||||||||||||||||||||||||
| Corporate debt securities | $ | $ | $ | ( | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
| Money market funds | |||||||||||||||||||||||||||||||||||||||||||||||
| Agency bonds | |||||||||||||||||||||||||||||||||||||||||||||||
| Treasury bills | |||||||||||||||||||||||||||||||||||||||||||||||
| Municipal debt securities | |||||||||||||||||||||||||||||||||||||||||||||||
| Total | $ | $ | $ | ( | $ | $ | $ | $ | ( | $ | |||||||||||||||||||||||||||||||||||||
Estimated fair values of available-for-sale securities are generally based on prices obtained from commercial pricing services. The following table summarizes the classification of the available-for-sale securities in the Company's Consolidated Balance Sheets (in thousands):
| June 30, 2025 | December 31, 2024 | ||||||||||
| Cash equivalents | $ | $ | |||||||||
| Marketable securities | |||||||||||
| Total | $ | $ | |||||||||
The following table summarizes the Company's portfolio of available-for-sale securities by contractual maturity (in thousands):
15
| June 30, 2025 | |||||||||||
| Amortized Cost | Estimated Fair Value | ||||||||||
| Less than one year | $ | $ | |||||||||
| Greater than one year | |||||||||||
| Total | $ | $ | |||||||||
Note 4 – Property and Equipment
Property and equipment consisted of the following (in thousands):
| June 30, 2025 | December 31, 2024 | ||||||||||
| Computer equipment | |||||||||||
| Leasehold improvements | |||||||||||
| Property and equipment, gross | |||||||||||
| Accumulated depreciation | ( | ( | |||||||||
| Property and equipment, net | $ | $ | |||||||||
The Company’s results included depreciation expense of approximately $52 thousand and $56 thousand for the six months ended June 30, 2025 and 2024, respectively.
Note 5 – Income (Loss) Per Share
For the six months ended June 30, 2025 and 2024, the Company incurred net losses and therefore no common stock equivalents were utilized in the calculation of diluted loss per share as they are anti-dilutive in the periods presented. At June 30, 2025 and 2024, the Company excluded the following potentially dilutive securities (in thousands):
| June 30, | |||||||||||
| 2025 | 2024 | ||||||||||
| Stock options | |||||||||||
| Warrants | |||||||||||
| Restricted stock units | |||||||||||
Note 6 – Fair Value Measurements
Fair value of financial assets and liabilities that are being measured and reported are defined as the exchange price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants in the principal market at the measurement date (exit price). The Company is required to classify fair value measurements in one of the following categories:
Level 1 inputs are defined as quoted prices (unadjusted) in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date.
Level 2 inputs are defined as inputs other than quoted prices included within Level 1 that are observable for the assets or liabilities, either directly or indirectly.
Level 3 inputs are defined as unobservable inputs for the assets or liabilities.
Financial assets and liabilities are classified based on the lowest level of input that is significant to the fair value measurement. The Company’s assessment of the significance of a particular input to the fair value measurement requires judgment and may affect the valuation of the fair value of assets and liabilities and their placement within the fair value hierarchy levels.
16
The Company's financial assets and liabilities that were accounted for at fair value on a recurring basis as of June 30, 2025 and December 31, 2024 were as follows (in thousands):
| June 30, 2025 | December 31, 2024 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Level 1 | Level 2 | Level 3 | Total | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||||||||||||||||||||||||||||||
| Assets: | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash equivalents | $ | $ | $ | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
| Marketable securities - available-for-sale | ||||||||||||||||||||||||||||||||||||||||||||||||||
| $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||
The carrying values of cash, cash equivalents, accounts receivable, accounts payable and accrued expenses approximate fair value as of June 30, 2025 and December 31, 2024, due to the short maturity nature of these items.
Note 7 – Accrued Liabilities
Accrued liabilities as of June 30, 2025 and December 31, 2024 were as follows (in thousands):
| June 30, 2025 | December 31, 2024 | ||||||||||
| Salaries, employee benefits and related taxes | $ | $ | |||||||||
| Clinical and R&D related liabilities | |||||||||||
| Accounting & tax consulting liabilities | |||||||||||
| Operating lease liabilities — current | |||||||||||
| Other | |||||||||||
| Total | $ | $ | |||||||||
Note 8 – Operating Leases
The Company has an operating lease for one office which expires on September 30, 2025. The Company estimates its incremental borrowing rate at lease commencement to determine the present value of lease payments as the Company's lease does not provide an implicit rate of return. The Company recognizes lease expense on a straight-line basis over the lease term. For lease agreements entered into or reassessed after the adoption of ASU No. 2016-02, Leases (Topic 842), the Company elected to account for non-lease components associated with its leases and lease components as a single lease component. The Company's lease includes an option for the Company to extend the lease term and/or sub-lease space in whole or in part.
17
Operating lease liabilities and right-of-use assets were recorded in the following captions of the Company's balance sheet as follows (in thousands):
| June 30, 2025 | December 31, 2024 | ||||||||||
| Right-of-Use Assets: | |||||||||||
| $ | $ | ||||||||||
| Total Right-of-Use Assets | $ | $ | |||||||||
| Operating Lease Liabilities: | |||||||||||
| $ | $ | ||||||||||
| Total Operating Lease Liabilities | $ | $ | |||||||||
As of June 30, 2025, the weighted average remaining lease term for the Company's operating lease was 0.25 years, and the weighted average discount rate for the Company's operating lease was 9.625 %. As of December 31, 2024, the weighted average remaining lease term for the Company's operating lease was 0.75 years, and the weighted average discount rate for the Company's operating lease was 9.625 %.
Future minimum lease payments under the lease agreement as of June 30, 2025 were as follows (in thousands):
| Years ended | Operating Leases | ||||
| 2025 | |||||
| Total lease payments | |||||
| Less: Amounts representing interest | ( | ||||
| Present value of lease liabilities | $ | ||||
Note 9 – Stockholders' Equity
Equity Issuances
At The Market Offering Agreement
On June 4, 2021, the Company entered into an At The Market Offering Agreement (the “ATM Agreement”) with H.C. Wainwright & Co., LLC, as sales agent, in connection with an “at the market offering” under which the Company from time to time may offer and sell shares of its common stock, having an aggregate offering price of up to $50.0 million. As of the date of this filing and so long as the Company’s public float remains below $75.0 million, the Company is subject to limitations pursuant to General Instruction I.B.6 of Form S-3 (the “Baby Shelf Limitation”), which limits the amount the Company can offer to up to one-third of its public float during any trailing 12-month period. Subsequent to the filing of a prospectus supplement to the Company's Registration Statement on Form S-3 (File No. 333-279034) relating to the at the market offering on August 21, 2024, the aggregate market value of its outstanding common stock held by non-affiliates was approximately $29.6 million. Pursuant to the Baby Shelf Limitation, since the aggregate market value of the Company's outstanding common stock held by non-affiliates was below $75.0 million at the time of such prospectus supplement filing, the aggregate amount of securities that the Company is permitted to offer and sell is now $9,855,890 , which is equal to one-third of the aggregate market value of our common stock held by non-affiliates as of August 20, 2024. If the Company’s public float exceeds $75.0 million on a future measurement date, it will no longer be subject to the Baby Shelf Limitation. During the six months ended June 30, 2025, the Company issued 68,496 shares of common stock under the ATM Agreement for net proceeds of $247,496 . Since inception through June 30, 2025, the Company has issued 136,669 shares of common stock under the ATM Agreement for net proceeds of $528,343 .
18
Stock Options and Warrants
| Stock Options | Warrants | |||||||||||||||||||||||||||||||||||||||||||||||||
| Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term (Years) | Aggregate Intrinsic Value (In Thousands) | Shares | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term (Years) | Aggregate Intrinsic Value (In Thousands) | |||||||||||||||||||||||||||||||||||||||||||
| Outstanding at December 31, 2024 | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Changes during the period: | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Granted | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercised | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Forfeited | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Expired | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Outstanding at June 30, 2025 | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Vested at June 30, 2025 or expected to vest in the future | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Vested at June 30, 2025 | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||
Restricted Stock
During the six months ended June 30, 2025 and 2024, the Company issued restricted stock for services as follows (in thousands, except share data):
| Six Months Ended June 30, | ||||||||||||||
| 2025 | 2024 | |||||||||||||
| Number of shares of restricted stock issued | ||||||||||||||
| Value of restricted stock issued | $ | $ | ||||||||||||
The weighted average estimated fair value of restricted stock issued for services in the six months ended June 30, 2025 and 2024 was $3.80 and $3.08 per share, respectively. The fair value of the restricted stock was determined using the Company’s closing stock price on the date of issuance. The vesting terms of restricted stock issuances are generally between one year and four years .
The following is a summary of the changes in non-vested restricted stock for the six months ended June 30, 2025:
| Restricted Stock Shares | Weighted Average Grant-Date Fair Value | ||||||||||
| Non-vested at December 31, 2024 | $ | ||||||||||
| Changes during the Year: | |||||||||||
| Granted | $ | ||||||||||
| Vested | ( | $ | |||||||||
| Forfeited | ( | $ | |||||||||
| Non-vested at June 30, 2025 | $ | ||||||||||
19
Restricted Stock Units
During the six months ended June 30, 2025 and 2024, the Company issued restricted stock units for services as follows (in thousands, except share data):
| Six Months Ended June 30, | |||||||||||
| 2025 | 2024 | ||||||||||
| Number of restricted stock units issued | |||||||||||
| Value of restricted stock units issued | $ | $ | |||||||||
The weighted average estimated fair value of restricted stock units issued for services in the six months ended June 30, 2025 and 2024 was $3.80 and $3.08 per share, respectively. The fair value of the restricted stock units was determined using the Company’s closing stock price on the date of issuance. The vesting terms of restricted stock unit issuances are generally one year , or upon the achievement of performance-based milestones.
The following is a summary of the changes in non-vested restricted stock units for the six months ended June 30, 2025:
| Restricted Stock Units | Weighted Average Grant-Date Fair Value | ||||||||||
| Non-vested at December 31, 2024 | $ | ||||||||||
| Changes during the Year: | |||||||||||
| Granted | $ | ||||||||||
| Vested | ( | $ | |||||||||
| Forfeited | $ | ||||||||||
| Non-vested at June 30, 2025 | $ | ||||||||||
Note 10 – Share-Based Compensation
Share-Based Compensation
The Company utilizes share-based compensation in the form of stock options, restricted stock, restricted stock units and warrants. The following table summarizes the components of share-based compensation expense for the three and six months ended June 30, 2025 and 2024 (in thousands):
| Three Months Ended June 30, | Six Months Ended June 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| Research and development | $ | $ | $ | $ | |||||||||||||||||||
| General and administrative | |||||||||||||||||||||||
| Total share-based compensation expense | $ | $ | $ | $ | |||||||||||||||||||
Total compensation cost related to unvested awards not yet recognized and the weighted-average periods over which the awards were expected to be recognized at June 30, 2025 were as follows (in thousands):
| Stock Options | Restricted Stock Units | Restricted Stock | |||||||||||||||
| Unrecognized compensation cost | $ | $ | $ | ||||||||||||||
| Expected weighted-average period in years of compensation cost to be recognized | |||||||||||||||||
20
Total fair value of shares vested and the weighted average estimated fair values of shares granted for the six months ended June 30, 2025 and 2024 were as follows (in thousands):
| Stock Options | Warrants | ||||||||||||||||||||||
| Six Months Ended June 30, | Six Months Ended June 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| Total fair value of shares vested | $ | $ | $ | $ | |||||||||||||||||||
| Weighted average estimated fair value of shares granted | $ | $ | $ | $ | |||||||||||||||||||
Valuation Assumptions
The fair value of stock options and warrants at the date of grant was estimated using the Black-Scholes option pricing model. The expected volatility is based upon historical volatility of the Company’s stock. The expected term for the options is based upon observation of actual time elapsed between date of grant and exercise of options for all employees. The expected term for the warrants is based upon the contractual term of the warrants.
Note 11 – Income Taxes
In assessing the realizability of deferred tax assets, including the net operating loss carryforwards (NOLs), the Company assesses the available positive and negative evidence to estimate if sufficient future taxable income will be generated to utilize its existing deferred tax assets. Based on its assessment, the Company has provided a full valuation allowance against its net deferred tax assets as their future utilization remains uncertain at this time.
As of December 31, 2024 and 2023, the Company had approximately $57.9 million and $43.7 million, respectively, of Federal NOLs available to offset future taxable income expiring from 2030 through 2036. The Company performed an analysis and determined that they had an ownership change of greater than 50% on September 15, 2022. As a result of the ownership change, $88.2 million of Federal NOLs will expire unutilized. The Company wrote off that portion of the deferred tax asset and reduced the corresponding valuation allowance resulting in $34.0 million of remaining Federal NOLs as of December 31, 2022. The write-off of the deferred tax asset and the corresponding reduction in valuation allowance has no impact to the consolidated balance sheet or income statement. Losses incurred before the ownership change on September 15, 2022 will be subject to an annual limitation of zero while losses incurred after September 15, 2022 will not be subject to limitations.
As of December 31, 2022, Cend Therapeutics, Inc. (“Cend”) had approximately $3.6 million of Federal NOLs available to offset future taxable income. The Company performed an analysis and determined that there was an ownership change of greater than 50% on September 15, 2022. As of September 15, 2022 Cend has approximately $3.1 million of Federal and $4.3 million of state NOLs. The state NOLs will expire from the 2036 through 2042 tax years. Using a fair market value of $36.1 million and applying an applicable federal rate of 2.54 % Cend will have an annual limitation of approximately $917 thousand each year. The Federal NOL of $459 thousand incurred in the post-acquisition period September 15, 2022 to December 31, 2022 is not subject to limitation, and does not expire.
As of December 31, 2024 and 2023, the Company's wholly owned Australian subsidiary had approximately $2.3 million and $2.4 million, respectively, of NOLs which will be carried forward and do not expire. There is a full valuation allowance against the NOLs.
As of December 31, 2024, the Company had federal research and development credit carryforwards of $0.5 million expiring from 2027 through 2034 if unutilized, and state research and development credit carryforwards of $0.1 million, which carryforward indefinitely. Utilization of these credits may be subject to an annual limitation based on changes in ownership.
As of December 31, 2024 and 2023, the Company had State NOLs available in New Jersey of $24.6 million and $19.4 million, respectively, California of $9.2 million and $9.2 million, respectively, and New York City of $1.9 million and $1.9 million, respectively, to offset future taxable income expiring from 2032 through 2044. The usage of the Company’s NOLs is limited given the change in ownership.
The Company applies the Financial Accounting Standards Board provisions for uncertain tax positions. The Company utilizes the two-step process to determine the amount of recognized tax benefit. For tax positions meeting the more-likely-than-not threshold, the amount recognized in the consolidated financial statements is the largest benefit that has a greater than 50% likelihood of being realized upon ultimate settlement with the relevant taxing authority. The Company recognizes interest and penalties associated with certain tax positions as a component of income tax expense.
21
As of December 31, 2024 and 2023, the Company’s uncertain tax positions were $344 thousand and $344 thousand, respectively. The uncertain tax positions are due to the acquisition of Cend related to Federal and state credits and certain state NOLs. The Company will continue to evaluate its uncertain tax positions in future periods. The Company does not believe there will be any material changes in its unrecognized tax positions over the next year.
For years prior to 2021, the federal statute of limitations is closed for assessing tax. The Company’s state tax returns remain open to examination for a period of three to four years from the date of the tax return filing.
In January 2025, the Company sold a portion of their unused New Jersey net operating losses through the State of New Jersey Economic Development Authority's (“NJEDA”) Technology Business Tax Certificate Transfer Program (“Program”). Under the Program, the Company sold $10.7 million of its New Jersey net operating losses (“NJ NOLs”) for net proceeds of $871 thousand. The sale of NJ NOLs resulted in a $962 thousand deferred income tax benefit and a loss on sale of $91 thousand recorded in other income (expense) in the consolidated financial statements.
Note 12 – Segment Information
The Company operates as one operating segment, the research and development of its investigational drug product. The Company used the management approach to determine its reportable operating segment. The Company's Chief Operating Decision Maker ("CODM") is its Chief Executive Officer, who reviews financial information presented on a consolidated basis. The Company is a clinical-stage pharmaceutical company and has limited revenue associated with a license and collaboration agreement. The CODM uses net loss as a measure of profit and loss, and assesses Company performance through the achievement of its clinical development goals. The CODM is regularly provided with budgeted and forecasted expense information which is used to determine the Company’s liquidity needs and cash allocation to its development programs. The CODM uses cash and marketable securities as a measure of segment assets in managing the enterprise.
The Company had revenue of $70 thousand and $70 thousand for the three and six months ended June 30, 2025, respectively, and $0 and $0 for the three and six months ended June 30, 2024, respectively. Depreciation and amortization expense was $44 thousand and $87 thousand for the three and six months ended June 30, 2025, respectively and $45 thousand and $91 thousand for the three and six months ended June 30, 2024, respectively. Equity method investment expense was $0 and $0 for the three and six months ended June 30, 2025, respectively, and $0 and $100 thousand for the three and six months ended June 30, 2024, respectively.
22
The following table illustrates our segment information for significant operating expenses and includes a reconciliation to net loss for the three and six months ended June 30, 2025 and 2024:
| Three Months Ended June 30, | Six Months Ended June 30, | ||||||||||||||||||||||
| 2025 | 2024 | 2025 | 2024 | ||||||||||||||||||||
| Revenue | $ | $ | $ | $ | |||||||||||||||||||
| Operating Expenses: | |||||||||||||||||||||||
| Research and development by significant expense: | |||||||||||||||||||||||
| BOLSTER trial | |||||||||||||||||||||||
| ASCEND trial | |||||||||||||||||||||||
| Chemistry, manufacturing and controls | |||||||||||||||||||||||
| Clinical department | |||||||||||||||||||||||
Other (1) | |||||||||||||||||||||||
| Research and development | |||||||||||||||||||||||
| General and administrative by significant expense: | |||||||||||||||||||||||
| Corporate | |||||||||||||||||||||||
| Investor relations/public relations/communications | |||||||||||||||||||||||
| Finance | |||||||||||||||||||||||
| Legal | |||||||||||||||||||||||
| Business development | |||||||||||||||||||||||
| Share based compensation expense | |||||||||||||||||||||||
Other (2) | |||||||||||||||||||||||
| General and administrative | |||||||||||||||||||||||
| Operating loss | ( | ( | ( | ( | |||||||||||||||||||
| Other income, net | |||||||||||||||||||||||
| Benefit from income taxes | ( | ( | |||||||||||||||||||||
| Net loss | $ | ( | $ | ( | $ | ( | $ | ( | |||||||||||||||
| Cash and marketable securities | $ | $ | |||||||||||||||||||||
(1) Included in Other are the GBM study, FORTIFIDE study and research oncology expenses | |||||||||||||||||||||||
(2) Included in Other are facilities expense, human resource and information technology expenses | |||||||||||||||||||||||
Note 13 – Australia Research and Development Tax Incentive
23
Note 14 – Commitments and Contingencies
Second Amended and Restated Employment Agreement
On June 10, 2025, the Company entered into an amended and restated employment agreement with David J. Mazzo, Ph.D., the Company’s Chief Executive Officer (the “Mazzo Second Amended and Restated Employment Agreement”). The Mazzo Second Amended and Restated Employment Agreement supplements and amends the Amended and Restated Employment Agreement, dated and effective as of March 19, 2021, between Dr. Mazzo and the Company.
The Mazzo Second Amended and Restated Employment Agreement: (i) amends Dr. Mazzo’s base salary to $717,229 from $633,032 and removed the non-accountable expense allowance of $12,000 per year, (ii) clarifies that a material reduction in Dr. Mazzo’s base salary is included in the definition of “Good Reason”, (iii) amends that in the event of Dr. Mazzo’s termination from the Company within two years following a Change in Control (as defined therein), any determination on (a) whether Dr. Mazzo’s termination constitutes a termination with or without Cause (as defined therein) or with or without Good Reason (as defined therein) and (b) Dr. Mazzo’s entitlement to the separation benefits and payments described therein, will be determined by the board of directors of the Company as constituted immediately prior to such Change in Control, which board may delegate its authority to a qualified independent third party, (iv) amends Dr. Mazzo’s COBRA benefit coverage after the date of termination to monthly reimbursement payments for the cost of Dr. Mazzo’s group health plan coverage, except in the event that such payment would incur tax penalties to the Company, violate applicable nondiscrimination requirements or in the event that the Company terminates the group health plan, in which case the Company will pay Dr. Mazzo a lump-sum cash payment equal to the aggregate cost of the remaining reimbursement payments less required withholdings, (v) amends the form of payment of Dr. Mazzo’s bonus payments in the event of a termination not in connection to a Change in Control to a lump-sum payment, (vi) amends the definition of “Change in Control” therein to include the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided that the Change in Control meets all of the requirements of a “change in control” within the meaning of Treasury Regulation §1.409A-3(i)(5), (vii) clarifies that the base salary severance payments, to the extent that the payments are in the aggregate less than or equal to twice the compensation limit under Section 401(a)(17) of the Internal Revenue Code of 1986, as amended (the “Code”), comply with the involuntary separation pay plan exception to Section 409A of the Code, and further clarifies that Dr. Mazzo may receive such severance payments as a lump sum payment on the next payroll period following the date of his termination from employment; and (viii) clarifies other technical terms for compliance with Section 409A of the Code.
Amended and Restated Employment Agreement
On June 10, 2025, the Company entered into an amended and restated employment agreement with Kristen K. Buck, M.D., the Company’s Vice President, R&D and Chief Medical Officer (the “Buck Amended and Restated Employment Agreement”). The Buck Amended and Restated Employment Agreement supplements and amends the Employment Agreement, dated and effective as of July 26, 2021, between Dr. Buck and the Company.
The Buck Amended and Restated Employment Agreement: (i) amends Dr. Buck’s base salary to $599,342 from $550,000 , (ii) clarifies that a material reduction in Dr. Buck’s base salary is included in the definition of “Good Reason”, (iii) amends that in the event of Dr. Buck’s termination from the Company within two years following a Change in Control (as defined therein), any determination on (a) whether Dr. Buck’s termination constitutes a termination with or without Cause (as defined therein) or with or without Good Reason (as defined therein) and (b) Dr. Buck’s entitlement to the separation benefits and payments described therein, will be determined by the board of directors of the Company as constituted immediately prior to such Change in Control, which board may delegate its authority to a qualified independent third party, (iv) amends Dr. Buck’s COBRA benefit coverage after the date of termination to monthly reimbursement payments for the cost of Dr. Buck group health plan coverage, except in the event that such payment would incur tax penalties to the Company, violate applicable nondiscrimination requirements or in the event that the Company terminates the group health plan, in which case the Company will pay Dr. Buck a lump-sum cash payment equal to the aggregate cost of the remaining reimbursement payments less required withholdings, (v) amends Dr. Buck’s bonus payments in the event of a termination not in connection to a Change in Control such that the amount will not be pro-rated for the number of days Dr. Buck was employed in the year of termination of employment, (vi) amends the definition of “Change in Control” therein to include the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided that the Change in Control meets all of the requirements of a “change in control” within the meaning of Treasury Regulation §1.409A-3(i)(5), (vii) clarifies that the base salary severance payments, to the extent that the payments are in the aggregate less than or equal to twice the compensation limit under Section 401(a)(17) of the Code comply with the involuntary separation pay plan exception to Section 409A of the Code, and further clarifies that Dr. Buck may receive such severance payments as a lump sum payment on the next payroll period following the date of her termination from employment; and (viii) clarifies other technical terms for compliance with Section 409A of the Code.
24
Amended and Restated Separation Benefits Agreement
On June 10, 2025, the Company entered into an amended and restated separation benefits agreement with James Nisco, Senior Vice President Finance and Treasury and Chief Accounting Officer of the Company (the “Nisco Amendment”). The Nisco Amendment supplements and amends the separation benefits provided in the Non-Change in Control Separation Pay Agreement between the Company and Mr. Nisco, dated as of December 24, 2024, and the severance agreement between Mr. Nisco and the Company, dated and effective as of September 12, 2016, and further amended on March 25, 2022.
The Nisco Amendment: (i) amends the definition of “Change in Control” therein to include the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided that the Change in Control meets all of the requirements of a “change in control” within the meaning of Treasury Regulation §1.409A-3(i)(5), (ii) includes a condition that Mr. Nisco’s right to the severance payments and benefits described therein are subject to Mr. Nisco’s timely execution and non-revocation of a general release of claims within sixty days following Mr. Nisco’s termination or such shorter time period set forth therein, (iii) amends Mr. Nisco’s COBRA benefit coverage after the date of termination to monthly reimbursement payments for the cost of Mr. Nisco’s group health plan coverage, except in the event that such payment would incur tax penalties to the Company, violate applicable nondiscrimination requirements or in the event that the Company terminates the group health plan, in which case the Company will pay Mr. Nisco a lump-sum cash payment equal to the aggregate cost of the remaining reimbursement payments less required withholdings, (iv) amends that in the event of Mr. Nisco’s termination from the Company within two years following a Change in Control (as defined therein), any determination on (a) whether Mr. Nisco’s termination constitutes a termination with or without Cause (as defined therein) or with or without Good Reason (as defined therein) and (b) Mr. Nisco’s entitlement to the separation benefits and payments described therein, will be determined by the board of directors of the Company as constituted immediately prior to such Change in Control, which board may delegate its authority to a qualified independent third party, (v) clarifies that the base salary severance payments, to the extent that the payments are in the aggregate less than or equal to twice the compensation limit under Section 401(a)(17) of the Code, comply with the involuntary separation pay plan exception to Section 409A of the Code, and further clarifies that Mr. Nisco may receive such severance payments as a lump sum payment on the next payroll period following the date of his termination from employment; and (vi) clarifies other technical terms for compliance with Section 409A of the Code.
Amended and Restated Separation Benefits Agreement
On June 10, 2025, the Company entered into an amended and restated separation benefits agreement (the “Imam Amendment”) with Tariq Imam, Senior Vice President, Business Development and Operations and General Counsel of the Company. The Imam Amendment supplements and amends the separation benefits provided in the Non-Change in Control Separation Pay Agreement between the Company and Mr. Imam, dated as of December 24, 2024, effective as of February 1, 2025 (the “Imam Non-Change in Control Separation Pay Agreement”) and the severance agreement between Mr. Imam and the Company, dated as of March 25, 2022 (the “Imam Severance Agreement”).
Imam Severance Agreement
Pursuant to the Imam Severance Agreement, Mr. Imam is entitled to receive certain payments following his termination from the Company in specific circumstances. If Mr. Imam terminates his employment for Good Reason (as defined in the Imam Severance Agreement) during the period commencing on the effective date of a Change in Control (as defined in the Imam Severance Agreement) and ending on the second anniversary of the effective date of a Change in Control (as defined in the Imam Severance Agreement), or the Company terminates Mr. Imam’s employment without Cause (as defined in the Imam Severance Agreement) (other than by reason of his death or disability), the Company will (a) continue to pay Mr. Imam’s current base salary of $356,000 (the “Imam Salary Payment”) for 12 months following the date the termination becomes effective (the "Imam Severance Period"), commencing on the next payroll period following the date the termination becomes effective and (b) pay Mr. Imam a lump-sum equal to 100 % of Mr. Imam’s then annual target bonus on the next payroll period following the date the termination becomes effective (the “Imam Cash Severance”) and (c) pay monthly the monthly premium amount for continued COBRA coverage. If the Company may not pay Mr. Imam’s COBRA premiums without incurring tax penalties or violating any requirement of law, the Company shall use its commercially reasonable best efforts to provide Mr. Imam with substantially similar assistance in an alternative manner (the “Imam COBRA Payments”), provided that the cost of doing so does not exceed the cost that the Company would have incurred had the Company been able to pay the COBRA premiums on Mr. Imam’s behalf. In addition to the Imam Salary Payment, Imam Cash Severance and the Imam COBRA Payments, the Company shall (i) fully vest as of the date the termination becomes effective, all outstanding unvested equity awards, including options granted to Mr. Imam; and (ii) extend the exercise date of the options to the earlier of the one-year anniversary of the date the termination becomes effective and the original expiration date of the options.
25
Imam Non-Change in Control Separation Pay Agreement
Pursuant to the Imam Non-Change in Control Separation Pay Agreement, Mr. Imam is entitled to a salary continuation of his current base salary of $356,000 for 12 months from the date of his termination, as well as a lump-sum cash payment of 100 % of his then annual target bonus on the next payroll period following the date the termination becomes effective. Mr. Imam is also entitled to payment of accrued but unpaid paid time off and wages up to the date of termination and monthly COBRA coverage reimbursements for 12 months from the date of termination. The Imam Non-Change in Control Separation Pay Agreement also provides an extension of the time period for exercising any fully vested option awards to the earlier of the one-year anniversary of the date the termination becomes effective and the original expiration date of the options.
The Imam Amendment: (i) amends the definition of “Change in Control” therein to include the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided that the Change in Control meets all of the requirements of a “change in control” within the meaning of Treasury Regulation §1.409A-3(i)(5), (ii) includes a condition that Mr. Imam’s right to the severance payments and benefits described therein are subject to Mr. Imam’s timely execution and non-revocation of a general release of claims within sixty days following Mr. Imam’s termination or such shorter time period set forth therein, (iii) amends Mr. Imam’s COBRA benefit coverage after the date of termination to monthly reimbursement payments for the cost of Mr. Imam’s group health plan coverage, except in the event that such payment would incur tax penalties to the Company, violate applicable nondiscrimination requirements or in the event that the Company terminates the group health plan, in which case the Company will pay Mr. Imam a lump-sum cash payment equal to the aggregate cost of the remaining reimbursement payments less required withholdings, (iv) clarifies that in the event of Mr. Imam’s termination from the Company within two years following a Change in Control (as defined therein), any determination on (a) whether Mr. Imam’s termination constitutes a termination with or without Cause (as defined therein) or with or without Good Reason (as defined therein) and (b) Mr. Imam’s entitlement to the separation benefits and payments described therein, will be determined by the board of directors of the Company as constituted immediately prior to such Change in Control, which board may delegate its authority to a qualified independent third party, (v) clarifies that the base salary severance payments, to the extent that the payments are in the aggregate less than or equal to twice the compensation limit under Section 401(a)(17) of Code, comply with the involuntary separation pay plan exception to Section 409A of the Code, and further clarifies that Mr. Imam may receive such severance payments as a lump sum payment on the next payroll period following the date of his termination from employment and (vi) clarifies other technical terms for compliance with Section 409A of the Code.
Legal Proceedings and Claims
From time to time, the Company is subject to legal proceedings and claims, either asserted or unasserted, that arise in the ordinary course of business. While the outcome of pending claims cannot be predicted with certainty, the Company does not believe that the outcome of any pending claims will have a material adverse effect on the Company's financial condition or operating results. The Company has elected to recognize expense for legal fees as incurred when the legal services are provided.
Note 15 – Technology Transfer Agreement
Impilo Therapeutics
In July 2023, the Company entered into a technology transfer agreement with Impilo Therapeutics (“Impilo”) under which the Company transferred its rights to its tumor penetrating nanocomplex (TPN) platform to Impilo. As consideration for the technology transfer, Impilo issued a total of 766,000 shares of its pre-seed preferred stock to the Company. On October 3, 2023, in connection with the Sanford Burnham Prebys license agreement (see Note 16 - License Agreements) Impilo cancelled the original stock certificate for 766,000 shares and reissued 574,500 shares of its pre-seed preferred stock to the Company.
26
On March 15, 2024, the Company purchased a Simple Agreement for Future Equity ("SAFE") from Impilo for $100 thousand. On July 12, 2024, the Company purchased an additional SAFE from Impilo for $30 thousand. As of June 30, 2025 and December 31, 2024, the Company owned 38.6 30.0 million and an 80 % discount rate.
Note 16 – License Agreements
Sanford Burnham Prebys
In December 2015, Cend entered into a license agreement with Sanford Burnham Prebys (“SBP”) under which Cend was granted an exclusive, worldwide, royalty-bearing license to certain patent rights and know-how controlled by SBP related to the development of certepetide. At the time the license agreement was entered into, Cend’s founding shareholder was an executive at SBP. The agreement provides the Company with the rights to grant and authorize sublicenses to use, sell, and otherwise exploit the patent rights. As consideration for the license, Cend issued a total of 382,030 shares of common stock, as adjusted for the Reverse Stock Split and Exchange Ratio. The Company is required to pay an annual license maintenance fee of $10 thousand increasing to $20 thousand on year seven of the agreement. The Company could also be required to make milestone payments to SBP upon completion of certain regulatory and commercial milestones. The aggregate potential milestone payments are approximately $10.6 million. The Company has also agreed to pay SBP royalties of 4 % of net sales of products sold by the Company, or through a sublicense, subject to certain reductions. Additionally, the Company is obligated to pay SBP 25 % of any sublicensing income received, which, pursuant to the technology transfer agreement with Impilo, resulted in SBP receiving 191,500 shares of the Company’s pre-seed preferred stock in Impilo on October 3, 2023.
The agreement will expire upon the later of (i) the final abandonment of all pending patent applications within the licensed patents or (ii) the expiration of the last to expire patent within the licensed patents. The agreement may be terminated in its entirety by the Company at any time by giving SBP sixty days’ prior written notice. The agreement may be terminated in its entirety by SBP if the Company, at any time, defaults in the payment of any sum when due and fails to make such payment within thirty days after receipt of written notice. The agreement may be terminated in its entirety by either SBP or the Company (i) in the event of an uncured material breach by the other party, or (ii) in the event the other party (a) files for, or is involuntarily petitioned with, bankruptcy (other than dissolution or winding up for the purposes of reconstruction or amalgamation), (b) makes an assignment of all or substantially all of its assets for the benefit of creditors, or (c) has a receiver or trustee is appointed and is unable to secure a dismissal, stay or other suspension of such proceedings within thirty days. Upon termination of the agreement for any reason, all rights and obligations of the Company with respect to the patents and patent applications shall terminate and revert to SBP.
SBP did no t own shares of the Company’s common stock as of June 30, 2025.
Note 17 – Research Collaboration and License Agreements
Exclusive License and Collaboration Agreement - Qilu Pharmaceutical
In February 2021, Cend entered into an Exclusive License and Collaboration Agreement (the “Qilu Agreement”) in which Cend granted an exclusive license to Qilu for the development and commercialization of certepetide in the Territory (defined as the Greater Area of China including China, Macau, Hong Kong, and Taiwan). Under the terms of the agreement, Qilu is solely responsible for the development of certepetide in its Territory. In consideration for the license, Qilu made an upfront payment of $10.0 million to Cend, which was recognized as revenue by Cend prior to the Company's acquisition of Cend on September 15, 2022 (the “Cend Merger”). In addition, Cend received and recognized as revenue a $5.0 million development milestone prior to the Cend Merger. The Company is eligible to receive additional development and commercial milestone payments up to $96.0 million and $125.0 million, respectively, tiered royalties on net sales ranging from 10 % to 15 %, and tiered sublicensing revenues ranging from 12 % to 35 %.
On March 25, 2024, the Company entered into a settlement agreement with Lingmed whereby Lingmed is entitled to 5.0 % of any future milestone payments received by the Company under the Qilu Agreement. In addition, Lingmed is entitled to a sum of $250 thousand with respect to the first future milestone received by the Company.
Unless terminated early, the Qilu Agreement will continue in effect until the expiration of all Qilu payment obligations. Either party may terminate the Qilu Agreement if an undisputed material breach by the other party is not cured within a defined
27
period of time, or upon notice for insolvency-related events of the other party that are not discharged within a defined time period. Qilu may terminate the Qilu Agreement in its entirety, at any time with at least sixty days written notice. All rights and obligations of Qilu with respect to such licensed patents and patent applications would terminate simultaneously.
Exclusive License and Collaboration Agreement - Kuva Labs
In November 2024, the Company entered into an Exclusive License and Collaboration Agreement in which the Company granted an exclusive license to Kuva Labs, Inc. (Kuva) to explore the synergistic potential of the Company's certepetide as a targeting and delivery agent for Kuva’s NanoMark™ imaging technology in solid tumors. Under the Agreement, Kuva will assume full responsibility for research, development, and commercialization costs, while the Company will be responsible for supplying certepetide for additional consideration pursuant to a Clinical Supply Agreement. In consideration for the license, the Company recognized $1.0 million as revenue upon delivery of the license in November 2024. The Company is eligible to receive additional development and commercial milestone payments up to $1.5 million and $17.5 million, respectively, a 5.0 % percent royalty on net sales, and sublicensing revenues of 50 %.
Note 18 – Subsequent Events
On July 15, 2025, the Company announced that it has been issued a patent from the United States Patent and Trademark Office covering the composition of matter of certepetide, more specifically, around its chemical structure and pharmacokinetic properties. The granted patent, U.S. Patent No. 12,351,653, further expands the Company’s intellectual property estate around certepetide and will expire in March 2040.
28
ITEM 2. MANAGEMENT'S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following Management’s Discussion and Analysis of Financial Condition and Results of Operations contains forward-looking statements that are subject to significant risks and uncertainties. There is no guarantee that our clinical development programs will be successful or result in the necessary regulatory approvals. Our actual results could differ materially from those anticipated in these forward-looking statements as a result of various factors, including those set forth under “Cautionary Note Regarding Forward-Looking Statements” herein and under “Risk Factors” in our 2024 Form 10-K. The following discussion should be read in conjunction with our consolidated financial statements and related notes thereto included elsewhere in this Quarterly Report and in our 2024 Form 10-K.
Overview
We are a clinical-stage pharmaceutical company dedicated to the discovery, development, and commercialization of innovative therapies for the treatment of solid tumors and other major diseases. Our investigational product, certepetide (formerly known as LSTA1 or CEND-1), is designed to activate a novel uptake pathway that allows co-administered or tethered (i.e., molecularly bound) anti-cancer drugs to target and penetrate solid tumors more effectively. Certepetide actuates this active transport system in a tumor-specific manner, resulting in systemically co-administered anti-cancer drugs more efficiently penetrating and accumulating in the tumor, while normal tissues are expected to remain unaffected. Certepetide has also been shown to modify the tumor microenvironment (“TME”) by reducing T-regulatory cells and augmenting cytotoxic T cells, thereby making tumors more susceptible to immunotherapies while also inhibiting the metastatic cascade (i.e., the spread of cancer to other parts of the body). We, our collaborators and other researchers have amassed and continue to amass significant non-clinical data demonstrating enhanced delivery of a range of existing and emerging anti-cancer therapies, including chemotherapeutics, immunotherapies, and RNA-based therapeutics. To date, certepetide has also demonstrated favorable safety, tolerability and activity in completed and ongoing clinical trials designed to enhance delivery of standard-of-care chemotherapy for pancreatic cancer. We are exploring certepetide as a means to enable a variety of therapeutic modalities to treat a range of solid tumors more effectively. Currently, certepetide is the subject of several Phase 2 clinical studies being conducted globally in a variety of solid tumor types, including metastatic pancreatic ductal adenocarcinoma (mPDAC), cholangiocarcinoma, appendiceal cancer, colon cancer and glioblastoma multiforme in combination with a variety of anti-cancer regimens.
Our leadership team has amassed several decades of collective biopharmaceutical and pharmaceutical product development experience across a variety of therapeutic categories and at all stages of development from preclinical through to product registration and launch. Our goal is to develop and commercialize products that address important unmet medical needs.
Targeted Solid Tumor Penetration via CendR Active Transport
Many solid tumor cancers, including, for example, pancreatic ductal adenocarcinoma (“PDAC”) and cholangiocarcinoma, are surrounded by dense fibrotic tissue, known as the tumor stroma. This stroma often limits the penetration of anti-cancer therapies including chemotherapy into the tumor and thus limits their efficacy. Emerging immunotherapies, including but not limited to checkpoint inhibitors and adoptive cell therapies (e.g., chimeric antigen receptor T cells (CAR-Ts)), also face challenges in effectively treating solid tumors. Many tumors exhibit an immunosuppressive TME, which suppresses a patient's immune system and can thus limit the effectiveness of immunotherapies and/or contribute to metastases. These factors, i.e., the combination of a dense stroma and an immunosuppressive TME, negatively impact the ability of many therapeutic agents to optimally treat these cancers.
To address the tumor stroma’s role as a key impediment to effective treatment, our approach is to activate the C-end rule (“CendR”), or CendR active transport mechanism, a naturally occurring transport system. Our investigational drug, certepetide (a specific, proprietary internalizing R-G-D or iRGD peptide), activates this transport system in a tumor-specific manner (Sugahara, Science, 2010). Certepetide enables more selective and efficient uptake of systemically administered anti-cancer drugs resulting in more intratumoral drug accumulation. The overall expected result is enhanced anticancer activity without an increase in systemic adverse side effects. While it is possible to couple/tether or conjugate some anticancer drugs to certepetide, we believe that our initial approach of co-administration of certepetide with anti-cancer therapies is advantageous. Co-administration does not create a new chemical entity (“NCE”) with its attendant development and regulatory hurdles, thereby providing an anticipated faster-to-clinic and faster-to-market product opportunity for a range of combination therapies. That said, an attractive life-cycle management strategy for certepetide would be to molecularly bind it to a variety of anti-cancer agents (as an alternative to co-administration), thereby creating new NCEs with the potential for distinct patent protection, compositionally or otherwise.
Certepetide has demonstrated favorable safety, tolerability, and activity to date in clinical trials enhancing the selective delivery of standard-of-care chemotherapies for mPDAC. Certepetide’s cancer targeting characteristics may also enable
29
emerging solid tumor treatment modalities to prove more effective. For example, preliminary results of certepetide in combination with both immunotherapy and chemotherapy are promising.
Certepetide as a treatment for solid tumor cancers in combination with other anti-cancer agents
Certepetide is an investigational drug that actuates the CendR active transport mechanism. Certepetide has been shown to modify the TME, making it less immunosuppressive and thereby making the tumor more susceptible to attack by the immune system while also inhibiting the metastatic cascade. It targets tumor vasculature, endothelial cells, tumor cells and some intratumoral immunosuppressive cells by its selective affinity for alpha-v beta-3 and alpha-v beta-5 integrins that are upregulated on these cells. Certepetide is a nine amino acid cyclic proprietary internalizing RGD (“iRGD”) peptide that, once bound to these integrins, undergoes proteolytic cleavage to release a linear peptide fragment, called a CendR peptide fragment. After dissociation from the integrin receptor, the CendR peptide fragment then binds with high selectivity and affinity to an adjacent receptor, called neuropilin-1, also upregulated in solid tumors, to activate the novel uptake pathway that allows circulating anticancer drugs to more selectively and effectively penetrate solid tumors. The ability of certepetide and iRGD peptides to modify the TME to enhance delivery and efficacy of co-administered drugs has been demonstrated in many preclinical models in a range of solid tumors. Lisata, its collaborators, and research groups around the world have published nearly 400 scientific papers related to the benefits of internalizing RGD peptides and the CendR pathway.
Clinically, certepetide has been the subject of multiple Phase 1 and Phase 2 trials. These recent studies include a Phase 1b/2a study in first line mPDAC patients. Study CEND1-201 was conducted in China by our development partner, Qilu Pharmaceutical. Two dose levels of certepetide (1.6 and 3.2 mg/kg) were combined with SoC chemotherapy (gemcitabine and nab-paclitaxel). There were 55 patients in the study, 53 of whom were evaluable for efficacy. Twenty-five (25) patients were treated with 1.6 mg/kg certepetide, and 28 patients with 3.2 mg/kg certepetide. In the 1.6 mg/kg certepetide group, partial response occurred in 11/25 (44.0%) patients, and stable disease occurred in 12/25 (48%) patients. In the 3.2 mg/kg certepetide group, partial response occurred in 11/28 (39.3%) patients, and stable disease occurred in 12/28 (42.9%) patients. The ORR was 41.5% for all doses. The ORR was 44.0% and 39.3% in 1.6 mg/kg group and in 3.2 mg/kg group, respectively. The DCR was 86.8% for all doses. The DCR in 1.6 and 3.2 mg/kg groups was 92.0% and 82.1%, respectively. The median PFS was 5.82 months for all doses combined. The median PFS was 7.36 months and 5.75 months in 1.6 mg/kg group and in 3.2 mg/kg group, respectively. The median OS was 11.10 months for all doses combined. The median OS was 10.35 months and 11.10 months in 1.6 mg/kg group and in 3.2 mg/kg group, respectively. The adverse event profile at both dose levels was similar to that for SoC alone. Qilu has completed Phase 2 enrollment (n=96) in first line mPDAC in combination with SoC gemcitabine and nab-paclitaxel.
Currently, certepetide is also the subject of a Phase 2 trial in first-line mPDAC patients, the ASCEND trial. ASCEND is being conducted in collaboration with the Australasian Gastrointestinal Clinical Trials Group (AGITG) and the University of Sydney at 25 sites in Australia and New Zealand. The Phase 2 double-blind, randomized (2:1), placebo-controlled, multi-center ASCEND trial is evaluating certepetide in combination with SoC chemotherapy (gemcitabine and nab-paclitaxel) for the treatment of mPDAC. The original ASCEND protocol included one dosing scheme for certepetide. Following the acquisition of Cend Therapeutics and, by extension, certepetide in September 2022, Lisata collaborated with AGITG to amend the protocol to ensure it respected international regulatory standards. Thus, endpoints typically recognized by regulators as primary in registration studies and more effective in guiding next stages of clinical development (e.g., overall survival), were added. The amended protocol was designed to assess the efficacy of two different dosing regimens of certepetide in two separate cohorts: Cohort A, with 95 patients receiving a single intravenous (IV) dose of certepetide 3.2 mg/kg or placebo in combination with SoC, and Cohort B, with 63 patients receiving two IV doses of certepetide 3.2 mg/kg or placebo administered 4 hours apart in combination with SoC. The preliminary data from Cohort A were reported at the ASCO GI meeting on January 24, 2025, demonstrating a median overall survival (mOS) of 12.68 months for the certepetide treated group, compared to 9.72 months for the placebo treated group. Despite a numerical trend in 6-month PFS favoring the certepetide treatment group, no significant improvement in median PFS was observed (mPFS of 5.5 months in both groups). However, the objective response rate (ORR) benefit is positive with 4/65 (6.2%) complete responses in the certepetide treated group, compared to 0/28 (0%) the placebo treated group. The preliminary data from Cohort B were presented at the ESMO-GI meeting in July 2025, demonstrating a six-month progression-free survival (“6MPFS”) of 60.8% for the certepetide-treated group, whereas the 6MPFS in the placebo-treated group was 25%. Median progression-free survival (“mPFS”) was 7.5 months for the certepetide-treated group and 4.7 months for the placebo-treated group. Objective response rate (“ORR”) was 45.2% for the certepetide-treated group and 19% for the placebo-treated group. Median overall survival (“mOS”) was 10.32 months for the certepetide-treated group compared to 9.23 months for the placebo-treated group. A comparison of the preliminary data from Cohorts A and B indicates that the addition of two doses of certepetide (Cohort B regimen) to SoC chemotherapy resulted in a clinically meaningful improvement in both PFS and ORR for patients with mPDAC. These clinically significant findings provide compelling support for the continued and expedited investigation of certepetide as a novel therapeutic agent for the treatment of metastatic pancreatic cancer. The adverse event profile of Cohorts A and B has been similar in subjects treated with certepetide compared to placebo,
30
confirming previous observations of certepetide’s benign safety profile. Final data and key findings from both cohorts of the ASCEND study are anticipated to be available later this year.
Additionally, certepetide is currently the subject of multiple ongoing and planned clinical trials being conducted globally in a variety of solid tumor types and in combination with several chemotherapy and immunotherapy anti-cancer regimens. The following diagram summarizes these studies.
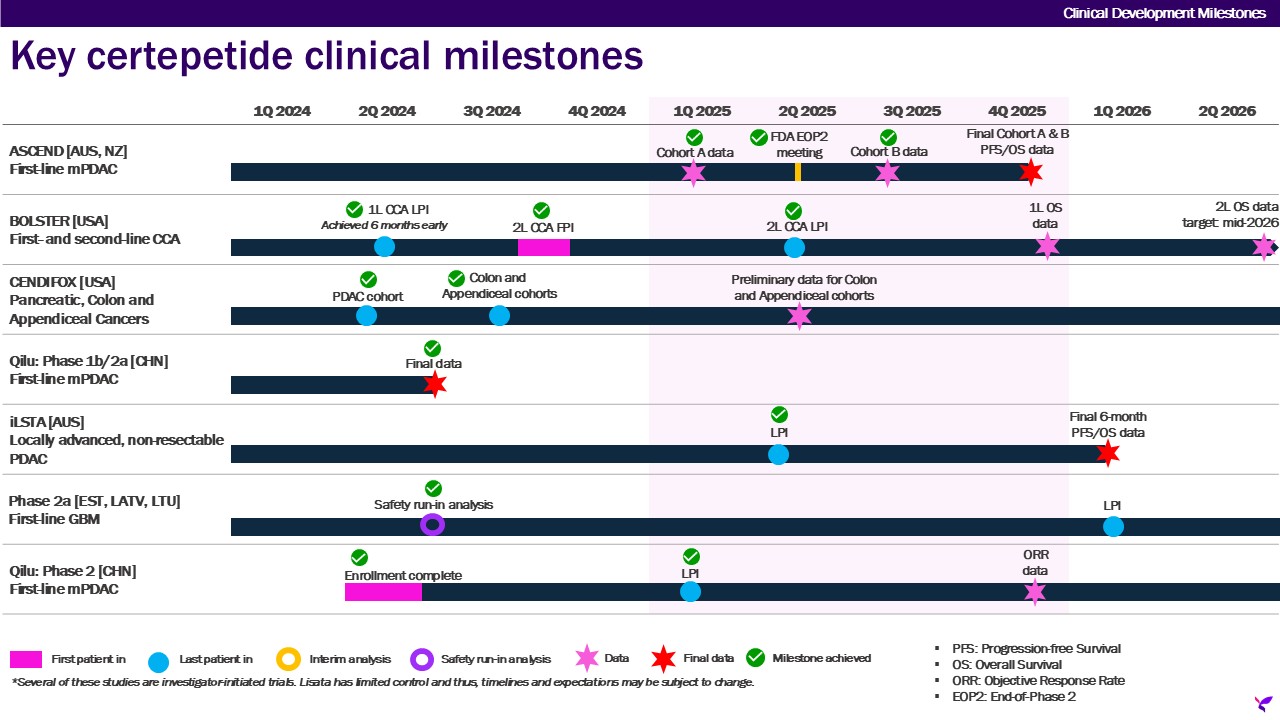
Additional Out-licensing Opportunities
Our intellectual property portfolio comprises notable programs available for out-licensing and/or partnering in order to augment or continue their clinical development. Our current long-term strategy focuses on advancing our therapies through development with the ultimate objective of obtaining market authorizations and entering commercialization, either alone or with partners, to provide treatment options to patients suffering from life-threatening medical conditions. We believe that we are well-positioned to realize potentially meaningful value increases within our own proprietary pipeline if we are successful in advancing our product candidates to their next significant development milestones.
31
Results of Operations
Three Months Ended June 30, 2025 Compared to Three Months Ended June 30, 2024
The following table summarizes our results of operations for the three months ended June 30, 2025 and June 30, 2024 (in thousands):
| Three Months Ended June 30, | |||||||||||||||||
| 2025 | 2024 | Change | |||||||||||||||
| Revenue | $ | 70 | $ | — | $ | 70 | |||||||||||
| Operating Expenses: | |||||||||||||||||
| Research and development | 2,253 | 2,601 | (348) | ||||||||||||||
| General and administrative | 2,685 | 2,922 | (237) | ||||||||||||||
| Total operating expenses | 4,938 | 5,523 | (585) | ||||||||||||||
| Loss from operations | (4,868) | (5,523) | 655 | ||||||||||||||
| Total other income | 209 | 479 | (270) | ||||||||||||||
| Benefit from income taxes | — | — | — | ||||||||||||||
| Net loss | $ | (4,659) | $ | (5,044) | $ | 385 | |||||||||||
Overall, net losses were $4.7 million for the three months ended June 30, 2025, compared to $5.0 million for the three months ended June 30, 2024.
Revenue
For the three months ended June 30, 2025, revenue totaled $70 thousand in connection with an upfront license fee related to a research license agreement. We did not have any revenue for the three months ended June 30, 2024.
Operating Expenses
For the three months ended June 30, 2025, operating expenses totaled $4.9 million, compared to $5.5 million for the three months ended June 30, 2024, representing a decrease of $0.6 million or 10.6%. Operating expenses are comprised of the following:
•Research and development expenses were approximately $2.3 million for the three months ended June 30, 2025, compared to $2.6 million for the three months ended June 30, 2024, representing a decrease of $0.3 million or 13.4%. This was primarily due to a reduction in patient treatment costs and clinical research organization (“CRO”) expenses associated with our Phase 2a proof-of-concept Bolster trial and lower spend on chemistry, manufacturing and controls (“CMC”).
•General and administrative expenses were approximately $2.7 million for the three months ended June 30, 2025, compared to $2.9 million for the three months ended June 30, 2024, representing a decrease of $0.2 million or 8.1%. This was primarily due to savings resulting from elimination of an employee position and lower spend on consulting and travel and entertainment expenses.
Historically, to minimize our use of cash, we have used a variety of equity instruments to compensate employees, consultants and other service providers. The use of these instruments has resulted in charges to the results of operations, which have been significant in the past.
32
Other Income (Expense)
Total other income (expense) is comprised primarily of investment income from cash, cash equivalents and marketable securities for the three months ended June 30, 2025 and 2024.
Six Months Ended June 30, 2025 Compared to Six Months Ended June 30, 2024
The following table summarizes our results of operations for the six months ended June 30, 2025 and June 30, 2024 (in thousands):
| Six Months Ended June 30, | |||||||||||||||||
| 2025 | 2024 | Change | |||||||||||||||
| Revenue | $ | 70 | $ | — | $ | 70 | |||||||||||
| Operating Expenses: | |||||||||||||||||
| Research and development | 4,856 | 5,842 | (986) | ||||||||||||||
| General and administrative | 5,930 | 6,282 | (352) | ||||||||||||||
| Total operating expenses | 10,786 | 12,124 | (1,338) | ||||||||||||||
| Loss from operations | (10,716) | (12,124) | 1,408 | ||||||||||||||
| Total other income | 371 | 881 | (510) | ||||||||||||||
| Benefit from income taxes | (962) | (798) | 164 | ||||||||||||||
| Net loss | $ | (9,383) | $ | (10,445) | $ | 1,062 | |||||||||||
Overall, net losses were $9.4 million for the six months ended June 30, 2025, compared to $10.4 million for the six months ended June 30, 2024.
Revenue
For the six months ended June 30, 2025, revenue totaled $70 thousand in connection with an upfront license fee related to a research license agreement. We did not have any revenue for the six months ended June 30, 2024.
Operating Expenses
For the six months ended June 30, 2025, operating expenses totaled $10.8 million, compared to $12.1 million for the six months ended June 30, 2024, representing a decrease of $1.3 million or 11.0%. Operating expenses are comprised of the following:
•Research and development expenses were approximately $4.9 million for the six months ended June 30, 2025, compared to $5.8 million for the six months ended June 30, 2024, representing a decrease of $1.0 million or 16.9%. This was primarily due to a reduction in clinical research organization (“CRO”) expenses, patient treatment costs and site expenses associated with our Phase 2a proof-of-concept Bolster trial and lower spend on chemistry, manufacturing and controls (“CMC”).
•General and administrative expenses were approximately $5.9 million for the six months ended June 30, 2025, compared to $6.3 million for the six months ended June 30, 2024, representing a decrease of $0.4 million or 5.6%. This was primarily due to one-off settlement costs in the prior year partially offset by severance costs in the current year.
Historically, to minimize our use of cash, we have used a variety of equity instruments to compensate employees, consultants and other service providers. The use of these instruments has resulted in charges to the results of operations, which have been significant in the past.
33
Other Income (Expense)
Total other income (expense) is comprised primarily of investment income from cash, cash equivalents and marketable securities and losses on sales of our New Jersey net operating losses for the six months ended June 30, 2025 and 2024.
Income Tax Benefit
In January 2025, we received final approval from the New Jersey Economic Development Authority (“NJEDA”) under the Technology Business Tax Certificate Transfer Program (the “Program”) to sell a percentage of our NJ NOLs, which were subsequently sold to a qualifying and approved buyer pursuant to the Program for net proceeds of $0.9 million. The $1.0 million of our NJ NOL tax benefits have been recorded as a benefit from income taxes and the loss on sale of $0.1 million recorded in other income (expense).
In March 2024, we received final approval from the NJEDA under the Program to sell a percentage of our NJ NOLs, which were subsequently sold to a qualifying and approved buyer pursuant to the Program for net proceeds of $0.7 million. The $0.8 million of NJ NOLs related tax benefits have been recorded as a benefit from income taxes and the loss on sale of $0.1 million recorded in other income (expense).
34
Analysis of Liquidity and Capital Resources
As of June 30, 2025, we had cash, cash equivalents and marketable securities of approximately $22.0 million, working capital of approximately $20.6 million, and stockholders’ equity of approximately $21.0 million.
During the six months ended June 30, 2025, we met our immediate cash requirements through existing cash balances. Additionally, we used equity and equity-linked instruments to pay for services and compensation.
Net cash (used in) or provided by, operating, investing and financing activities were as follows (in thousands):
| Six Months Ended June 30, | |||||||||||
| 2025 | 2024 | ||||||||||
| Net cash used in operating activities | $ | (9,358) | $ | (12,310) | |||||||
| Net cash provided by investing activities | 9,936 | 18,149 | |||||||||
| Net cash provided by (used in) financing activities | 41 | (110) | |||||||||
Operating Activities
Our cash used in operating activities during the six months ended June 30, 2025 was $9.4 million, which is comprised of (i) our net loss of $9.4 million, adjusted for non-cash expenses totaling $0.8 million (which includes adjustments for equity-based compensation, depreciation and amortization, and amortization/accretion of marketable securities) and (ii) changes in operating assets and liabilities using approximately $0.8 million.
Our cash used in operating activities during the six months ended June 30, 2024 was $12.3 million, which is comprised of (i) our net loss of $10.4 million, adjusted for non-cash expenses totaling $0.6 million (which includes adjustments for equity-based compensation, depreciation and amortization, loss from equity method investment, and amortization/accretion of marketable securities) and (ii) changes in operating assets and liabilities using approximately $2.5 million.
Investing Activities
Our cash provided by investing activities during the six months ended June 30, 2025 totaled $9.9 million and was primarily due to net sales of marketable securities (net of purchases of marketable securities).
Our cash provided by investing activities during the six months ended June 30, 2024 totaled $18.1 million and was primarily due to net sales of marketable securities (net of purchases of marketable securities) partially offset by an investment of $0.1 million in Impilo Therapeutics.
Financing Activities
Our cash provided by financing activities during the six months ended June 30, 2025 totaled $41 thousand and consisted of $0.3 million in proceeds primarily from the issuance of shares through our ATM Agreement (as defined below) partially offset by tax withholding-related payments on net share settlement equity awards to employees of $0.3 million.
Our cash used in financing activities during the six months ended June 30, 2024 totaled $0.1 million and consisted primarily of tax withholding-related payments on net share settlement equity awards to employees.
Liquidity and Capital Requirements Outlook
To meet our short and long-term liquidity needs, we expect to use existing cash balances, marketable securities and a variety of other means. Other sources of liquidity could include additional potential issuances of debt or equity securities in public or private financings, partnerships and/or collaborations and/or sale of assets. Our history of operating losses and liquidity challenges may make it difficult for us to raise capital on acceptable terms or at all. The demand for the equity and debt of pharmaceutical companies like ours is dependent upon many factors, including the general state of the financial markets. During times of extreme market volatility, capital may not be available on favorable terms, if at all. Our inability to obtain such additional capital could materially and adversely affect our business operations. We will also continue to seek, as appropriate, grants for scientific and clinical studies from various governmental agencies and foundations, and other sources of non-dilutive funding. We believe that our cash on hand and marketable securities will enable us to fund current operating expenses for at least the next 12 months following the issuance of our financial statements. Our future capital requirements are difficult to forecast and will depend on many factors including the timing and nature of any other strategic transactions that we undertake; and our ability to establish and maintain collaboration partnerships, in-license/out-license or other similar arrangements and the financial terms of such agreements.
35
On June 4, 2021, we entered into the ATM Agreement with H.C. Wainwright & Co., LLC as sales agent, in connection with an “at the market offering” under which we from time to time may offer and sell shares of our common stock having an aggregate offering price of up to $50.0 million. As of the date of this filing and so long as our public float remains below $75.0 million, we are subject to limitations pursuant to General Instruction I.B.6 of Form S-3 (the “Baby Shelf Limitation”), which limits the amount we can offer to up to one-third of our public float during any trailing 12-month period. Subsequent to the filing of a prospectus supplement to our Registration Statement on Form S-3 (File No. 333-279034) relating to the at the market offering on August 21, 2024, the aggregate market value of our outstanding common stock held by non-affiliates was approximately $29.6 million. Pursuant to the Baby Shelf Limitation, since the aggregate market value of our outstanding common stock held by non-affiliates was below $75.0 million at the time of such prospectus supplement filing, the aggregate amount of securities that we are permitted to offer and sell is now $9,855,890, which was equal to one-third of the aggregate market value of our common stock held by non-affiliates as of August 20, 2024. If our public float exceeds $75.0 million on a future measurement date, the Company will no longer be subject to the Baby Shelf Limitation. During the six months ended June 30, 2025, we issued 68,496 shares of common stock under the ATM Agreement for net proceeds of $247,496. Since inception through June 30, 2025, we have issued 136,669 shares of common stock under the ATM Agreement for net proceeds of $528,343.
While we continue to seek capital through a number of means, there can be no assurance that additional financing will be available on acceptable terms, if at all, and our negotiating position in capital generating efforts may worsen as existing resources are used. Additional equity financing may be dilutive to our stockholders; debt financing, if available, may involve significant cash payment obligations and covenants that restrict our ability to operate as a business; our stock price may not reach levels necessary to induce option or warrant exercises; and asset sales may not be possible on terms we consider acceptable. If we are unable to access capital necessary to meet our long-term liquidity needs, we may have to delay the expansion of our business or raise funds on terms that we currently consider unfavorable.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements.
Critical Accounting Policies and Estimates
There have been no material changes in our critical accounting policies and estimates during the six months ended June 30, 2025, compared to those reported in our 2024 Form 10-K.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
Not applicable.
ITEM 4. CONTROLS AND PROCEDURES
(a) Disclosure Controls and Procedures
Disclosure controls and procedures are our controls and other procedures that are designed to ensure that information required to be disclosed in the reports that we file or submit under the Securities Exchange Act of 1934 (as defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934), as amended (the “Exchange Act”) is recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms. Disclosure controls and procedures include, without limitation, controls and procedures designed to ensure that information required to be disclosed in the reports that we file under the Exchange Act is accumulated and communicated to management, including our Chief Executive Officer, who serves as our principal executive officer, and our Senior Vice President, Finance and Treasury and Chief Accounting Officer, who serves as our principal financial officer (together, the “Evaluating Officers”), as appropriate, to allow timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well-designed and operated, can provide only reasonable assurance of achieving the desired control objectives. Due to the inherent limitations of control systems, not all misstatements may be detected. These inherent limitations include the realities that judgments in decision-making can be faulty and that breakdowns can occur because of a simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the control. Controls and procedures can only provide reasonable, not absolute, assurance that the above objectives have been met.
36
As of June 30, 2025, we evaluated, with the participation of our management, including our Evaluating Officers, the effectiveness of our disclosure controls and procedures as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act. Based on that evaluation, our Evaluating Officers concluded that our disclosure controls and procedures were effective, at the reasonable assurance level, in ensuring that information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s rules and forms and is accumulated and communicated to management, including the Evaluating Officers, as appropriate to allow timely decisions regarding required disclosure.
(b) Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting, as such term is defined in Exchange Act Rule 13a-15, that occurred during our last quarter to which this Quarterly Report relates that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
37
PART II
OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
We are not currently a party to any material legal proceedings.
ITEM 1A. RISK FACTORS
There have been no material changes to the risk factors previously reported in our 2024 Form 10-K. See the risk factors set forth in our 2024 Annual Report on Form 10-K under the caption “Item 1A - Risk Factors.”
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS
None.
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
None.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
ITEM 5. OTHER INFORMATION
During the three months ended June 30, 2025, no director or officer of the Company adopted, modified or terminated a “Rule 10b5-1 trading arrangement” or “non-Rule 10b5-1 trading arrangement,” as each term is defined in Item 408(a) of Regulation S-K.
ITEM 6. EXHIBITS
The Exhibit Index appearing immediately after the signature page to this Form 10-Q is incorporated herein by reference.
38
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| LISATA THERAPEUTICS, INC. | ||||||||
| August 7, 2025 | By: /s/ David J. Mazzo, PhD Name: David J. Mazzo, PhD Title: President & Chief Executive Officer (Principal Executive Officer) | |||||||
| August 7, 2025 | By: /s/ James Nisco Name: James Nisco Title: SVP, Finance and Treasury and Chief Accounting Officer (Principal Financial Officer and Principal Accounting Officer) | |||||||
39
LISATA THERAPEUTICS, INC.
FORM 10-Q
Exhibit Index
| Second Amended and Restated Employment Agreement, by and between the Company and David J. Mazzo, Ph.D., dated as of June 10, 2025 (filed as Exhibit 10.1 to the Company's Current Report on 8-K, filed with the SEC on June 13, 2025) | ||||||||
| Amended and Restated Employment Agreement, by and between the Company and Kristen K. Buck, M.D., dated as of June 10, 2025 (filed as Exhibit 10.2 to the Company's Current Report on 8-K, filed with the SEC on June 13, 2025) | ||||||||
| Amended and Restated Separation Benefits Agreement, by and between the Company and James Nisco, dated as of June 10, 2025 (filed as Exhibit 10.3 to the Company's Current Report on 8-K, filed with the SEC on June 13, 2025) | ||||||||
| Amended and Restated Separation Benefits Agreement, by and between the Company and Tariq Imam, dated as of June 10, 2025 (filed as Exhibit 10.4 to the Company's Current Report on 8-K, filed with the SEC on June 13, 2025) | ||||||||
| * | Certification of Principal Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |||||||
| * | Certification of Principal Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act of 2002 | |||||||
| ** | Certification of Principal Executive Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |||||||
| ** | Certification of Principal Financial Officer pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 | |||||||
| 101.INS | Inline XBRL Instance Document - the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document | |||||||
| 101.SCH | Inline XBRL Taxonomy Extension Schema | |||||||
| 101.CAL | Inline XBRL Taxonomy Extension Calculation Linkbase | |||||||
| 101.DEF | Inline XBRL Taxonomy Extension Definition Linkbase | |||||||
| 101.LAB | Inline XBRL Taxonomy Extension Label Linkbase | |||||||
| 101.PRE | Inline XBRL Taxonomy Extension Presentation Linkbase | |||||||
| 104 | Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibit 101) | |||||||
_______________
| * | Filed herewith. | ||||
| ** | Furnished herewith. | ||||
40

Exhibit 10.1 SECOND AMENDED AND RESTATED EMPLOYMENT AGREEMENT This SECOND AMENDED AND RESTATED EMPLOYMENT AGREEMENT (this “Agreement”), dated as of June 10, 2025, and effective on June 10, 2025, (the “Effective Date”) is by and between Lisata Therapeutics, Inc. (the “Company”) and David J. Mazzo, Ph.D. (the “Executive”). WITNESSETH: WHEREAS, the Company desires to continue to employ the Executive as its Chief Executive Officer and the Executive desires to be so employed by the Company; and WHEREAS, the Company and the Executive each believe it is in their respective best interests to enter into this Agreement setting forth the mutual understandings and agreements reached between the Company and the Executive with respect to the Executive’s employment with the Company and certain restrictions on the Executive’s conduct benefitting the Company during such time and thereafter, all as set forth herein. NOW, THEREFORE, in consideration of the promises and the mutual covenants and agreements contained herein and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto, intending to be legally bound hereby, agree as follows: Section 1. Employment. The Company agrees to employ the Executive, and the Executive agrees to be employed by the Company on a full-time basis, for the period commencing on the Effective Date and, subject to earlier termination pursuant to Section 6 below, continuing until December 31, 2025 (the “Initial Term”). Unless Executive is given written notice by the Company (acting through the Company’s Board of Directors (the “Board”)) no later than ninety (90) days prior to the expiration of the Initial Term, Executive’s employment hereunder shall be deemed extended for an additional period of one (1) year, subject to earlier termination pursuant to Section 6 below (each, a “Renewal Term”), in each such case, commencing upon the expiration of the Initial Term or the then-current Renewal Term, as the case may be. As used in this Agreement, the “Term” shall refer to the period beginning on the Effective Date and ending on the effective date of the termination of this Agreement and the Executive’s employment hereunder (the “Termination Date”) in accordance with this Section 1 or Section 6 below. The Executive hereby represents and warrants to the Company that he has the legal capacity to execute and perform this Agreement, and that its execution and performance by him will not violate the terms of any existing agreement or understanding to which the Executive is a party; and the Company hereby represents and warrants to the Executive that the person executing this Agreement on its behalf has the authority to do so and to bind the Company. Section 2. Position and Duties. During the Term, the Executive shall be employed as the Company’s President and Chief Executive Officer and shall perform duties consistent with such position, including but not limited to direct responsibility for all day-to-day operations of the Company, and such other related duties as the Board shall reasonably request. The Executive will

2 report to the Chairman of the Board, and be subject to the lawful direction of, the Board. During the Term, and except for PTO in accordance with Section 5(a) below, the Executive shall devote his full business time, attention, skill and efforts to the business and affairs of the Company, its subsidiaries and other affiliates and shall comply with the Company’s codes of conduct, policies and procedures as applicable at any given time; provided however; the foregoing shall not prevent the Executive from (a) engaging in not-for-profit activities (e.g., board membership with charitable, educational, or religious organizations), (b) serving on the board of directors (or similar governing body), subject to the prior written approval of the Board, which shall not be unreasonably withheld, of not more than two (2) other public corporations (or other business entities) that are not competitors of the Company (as determined in good faith by the Board, it being understood that a failure to approve if service would be inconsistent with ISS standards is reasonable), and (c) managing the Executive’s personal and immediate family member’s passive investments, as long as, in each case, such activities individually or in the aggregate do not materially interfere or conflict with the Executive’s duties hereunder or create a potential business or fiduciary conflict (in each case, as determined in good faith by the Board). During the Term, the Executive also shall serve as a member of the Board and in such other executive-level positions or capacities as may, from time to time, be reasonably requested by the Chairman and/or the Board, including, without limitation (subject to election, appointment, re-election or re-appointment, as applicable) as (i) a member of the board of directors or similar governing body of any of the Company’s subsidiaries or other affiliates, (ii) an officer of any of the Company’s subsidiaries or other affiliates, and/or (iii) a member of any committee of the Company and/or any of its subsidiaries or other affiliates, in each case, for no additional compensation. The Executive’s position as a member of the Board shall be subject to election and/or re-election by the Company’s shareholders, and the failure of the Company’s shareholders to elect or re-elect the Executive shall not be deemed either (A) a breach by the Company of its obligation to the Executive or (B) a material reduction in the Executive’s position, duties, responsibilities or authority. The Executive’s principal place of employment for the performance of his services hereunder shall be at the Company’s corporate headquarters in Basking Ridge, NJ; provided; however, it is understood that the Executive shall be required to travel (both within the US and abroad) as reasonably necessary to perform his duties hereunder. Section 3. Compensation. For all services rendered by the Executive in any capacity required hereunder during the Term, the Executive shall be compensated as follows: (a) The Company shall pay the Executive a base salary (the “Base Salary”) at the annualized rate of $717,229.00, which shall be subject to customary withholdings and authorized deductions and be payable in equal installments in accordance with the Company’s customary payroll practices in place from time to time. The Executive’s Base Salary shall be subject to review by the Board and/or the Compensation Committee of the Board (the “Compensation Committee”) at least annually and may be increased, but not decreased, from time to time by the Board. As used in this Agreement, the term “Base Salary” shall refer to base salary as may be adjusted from time to time. (b) The Executive shall be entitled to participate in all compensation and employee benefit plans or programs and to receive all other benefits and perquisites that are approved by the Board and are generally made available by the Company to other senior executives of the Company and to the extent permissible under the general terms and provisions of such plans

3 or programs and in accordance with the provisions thereof. Notwithstanding any of the foregoing, nothing in this Agreement shall require the Company or any subsidiary or affiliate thereof to establish, maintain or continue any particular plan or program nor preclude the amendment, rescission or termination of any such plan or program that may be established from time to time. (c) The Executive is eligible to receive an annual equity award, the size and value of which would be determined by the Board (or the Compensation Committee of the Board) in its discretion. Any equity award will be granted in accordance with the terms of the applicable equity plan or arrangement that may be in effect from time to time. (d) The Executive shall be eligible to receive an annual cash bonus for each calendar year ending during the Term (“Annual Bonus”). The Executive’s target Annual Bonus will equal 55% of his Base Salary (the “Target Bonus”) and the Executive’s maximum Annual Bonus will equal 100% of his Base Salary. Annual Bonus will be determined by the Board and/or the Compensation Committee based upon the level of achievement of the Company’s corporate goals and objectives for the calendar year with respect to which the Annual Bonus relates and the Executive’s individual performance (in each case, as reasonably determined by the Board and/or the Compensation Committee). The Board agrees to take into account the Executive’s input with respect to the establishment of the Executive’s individual goals and objectives. The Board may, in its discretion, award the Executive an Annual Bonus of less than or greater than the Target Bonus depending on the Executive’s level of achievement of the performance targets. Each Annual Bonus for a calendar year, to the extent earned, will be paid in a lump sum in the following calendar year, but no later than March 15 of the calendar year immediately following the applicable calendar year for which the Annual Bonus is being measured. In order for the Executive to receive an Annual Bonus, the Executive must be actively employed by the Company on December 31 of the calendar year for which the Annual Bonus is being measured. Section 4. Business Expenses. The Company shall pay or reimburse the Executive for all reasonable travel and other reasonable expenses incurred by the Executive in connection with the performance of his duties and obligations under this Agreement, subject to the Executive’s presentation of appropriate vouchers or receipts in accordance with such policies and approval procedures as the Company may from time to time establish for employees (including but not limited to prior approval of extraordinary expenses) and to preserve any deductions for Federal income taxation purposes to which the Company may be entitled. Section 5. Benefits; Perquisites; Expense Reimbursement. In addition to those payments and benefits set forth above or elsewhere herein, the Executive shall be entitled to the following other benefits and payments: (a) Vacation. Executive shall be entitled to twenty-nine (29) days of paid time off (“PTO”) per calendar year (prorated in the event of a service year which is shorter than a calendar year) under the Company’s current PTO Policy, in addition to Company-observed holidays. Any PTO time not used during a calendar year shall be treated in accordance with the Company’s policies relating to unused PTO time.

4 (b) Supplemental Term Life and Long-Term Disability Insurance. The Company will pay up to $10,000 annually (in the aggregate) for supplemental term life insurance coverage and supplemental long-term disability coverage. (c) D&O Insurance. The Executive shall be covered by the Directors and Officers Liability Insurance policy that generally covers the directors and officers of the Company, provided by the Company at its expense. (d) Indemnification. The Executive shall be entitled to the benefit of the indemnification provisions contained in the Company’s By-Laws or Certificate of Incorporation as they may be amended from time to time, to the extent permitted by applicable law, at the time of the assertion of any liability against the Executive. Section 6. Termination of Employment. (a) Events of Termination. The Executive’s employment hereunder may be terminated upon the occurrence of any of the following events: (i) Termination for Cause. The Company (acting through the Board) may terminate the Executive’s employment hereunder for Cause at any time. For purposes of this Agreement, “Cause” shall mean that, as determined by the Board, the Executive has: (A) committed gross negligence in connection with his duties as set forth herein or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (B) committed fraud in connection with his duties as set forth herein or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (C) engaged in personal dishonesty, willful misconduct, willful violation of any law, or breach of fiduciary duty, in each instance, with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (D) been indicted for, or has been found by a court of competent jurisdiction to have committed or plead guilty to, (1) a felony (or state law equivalent) or (2) any other serious crime involving moral turpitude or that has (or is reasonably likely to have) a material adverse effect either on (x) the Executive’s ability to perform his duties under the Agreement or (y) the reputation and goodwill of the Company, regardless of whether or not such other crime is related or unrelated to the business of the Company, its subsidiaries or other affiliates; (E) shown chronic use of alcohol, drugs or other similar substances that materially affects the Executive’s work performance; (F) breached his obligations under (1) this Agreement, (2) the Confidentiality, Non- Compete and Inventions Assignment Agreement attached hereto as Exhibit A (the “Covenants Agreement”) or (3) any other agreement executed by the Executive for the benefit of the Company, its subsidiaries and/or other affiliates, provided, that, if such breach described in this clause (F) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to cure such breach; (G) failed to materially perform the Executive’s duties or to follow the lawful directives of the Board; provided, that, if such failure described in this clause (G) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to cure such failure; or (H) materially violated the Company’s written code of conduct or other written or established policies and/or procedures in place from time to time; provided, that, if such violation described in this clause (H) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to

5 cure such violation. Any notice to the Executive under this Section 6(a)(i) shall be in writing and shall specify in reasonable detail the Executive’s acts or omissions that the Company alleges constitute “Cause.” (ii) Termination without Cause. The Company (acting through the Board) may terminate the Executive’s employment hereunder without Cause (other than by reason of death or Disability) at any time upon notice to Executive. (iii) Resignation for Good Reason. The Executive may voluntarily terminate his employment hereunder for Good Reason (as defined below) upon written notice to the Company in accordance with the definition thereof. For purposes of this Agreement, “Good Reason” shall mean the occurrence of any of the following events: (A) material breach by the Company of its obligations under this Agreement; (B) the Executive’s base salary, position, duties, responsibilities, or authority have been materially reduced or the Executive has repeatedly been assigned duties that are materially inconsistent with his duties set forth herein, in each case, without the Executive’s consent or (C) the relocation of the Executive’s principal place of employment, without the Executive’s consent, in a manner that lengthens his one-way commute distance by fifty (50) or more miles. “Good Reason” shall not be deemed to exist, however, unless (1) the Executive shall have given written notice to the Company specifying in reasonable detail the Company’s acts or omissions that the Executive alleges constitute “Good Reason” within sixty (60) days after the first occurrence of such circumstances and the Company shall have failed to cure any such act or omission within sixty (60) days of receipt of such written notice, and (2) the Executive actually terminates employment within one hundred eighty (180) days following the initial occurrence of any of the foregoing conditions that he considers to be “Good Reason.” If the Executive fails to provide this notice and cure period prior to his resignation or resigns more than one hundred eighty (180) days after the initial existence of the condition, his resignation will not be deemed to be for “Good Reason.” (iv) Resignation without Good Reason. The Executive may voluntarily terminate his employment hereunder for any reason at any time, for any reason that does not constitute Good Reason, upon thirty (30) days’ prior written notice to the Company, provided, however, the Company reserves the right, upon written notice to the Executive, to accept the Executive’s notice of resignation and to accelerate such notice and make the Executive’s resignation effective immediately, or on such other date prior to the Executive’s intended last day of work as the Company deems appropriate. It is understood and agreed that the Company’s election to accelerate Executive’s notice of resignation shall not be deemed a termination by the Company without Cause for purposes of Section 6(a)(ii) of this Agreement, Section 7(a) of this Agreement or otherwise, or constitute Good Reason for purposes of Section 6(a)(iii) of this Agreement, Section 7(a) of this Agreement or otherwise. (v) Disability. The Executive’s employment hereunder shall terminate upon his Disability. For purposes of this Agreement, “Disability” shall mean that the Executive has been unable to perform his duties to the Company on account of physical or mental illness or incapacity for a period of ninety (90) consecutive calendar days or one hundred twenty (120) calendar days (whether or not consecutive) during any 365-day period, as a result of a condition that is treated as a total or permanent disability under the long-term disability insurance policy of the Company that covers the Executive.

6 (vi) Death. The Executive’s employment hereunder shall automatically terminate upon his death. (b) Resignation from Directorships, Officerships and Committees. The termination of the Executive’s employment for any reason shall constitute the Executive’s resignation from (i) any director, officer, employee or committee position the Executive has with the Company or any of its affiliates and (ii) all fiduciary positions the Executive holds with respect to any employee benefit plans or trusts established by the Company. The Executive agrees that this Agreement shall serve as written notice of resignation in this circumstance; provided, however, the Executive agrees to take any additional actions that are deemed reasonably necessary by the Company to effectuate or evidence such resignations. (c) Interpretation. For purposes of this Section 6, the Board will determine in its reasonable discretion whether a termination of Executive’s employment constitutes a termination with Cause under Section 6(a)(i), a termination without Cause under Section 6(a)(ii), a resignation with Good Reason under Section 6(a)(iii), or a resignation without Good Reason under Section 6(a)(iv). If the Company consummates a Change in Control, the members of the Board as constituted immediately prior to the closing of a Change in Control (the “Incumbent Board”) shall retain authority to make the forgoing determinations concerning the termination of Executive’s employment until the second (2nd) anniversary of the closing date of the Change in Control regardless of whether the Company’s Board is reconstituted in connection with the Change in Control. Following a Change in Control, the Incumbent Board may delegate its authority under this Section 6(c) to a qualified independent third party, such as a retired judge selected by the head of the American Arbitration Association in Manhattan, an independent compensation consultant or a law firm. Section 7. Compensation upon Termination of Employment. All defined terms used in this Section 7 but not defined in this Section 7 or elsewhere in this Agreement shall have the meanings ascribed to such terms in the Covenants Agreement: (a) Termination without Cause; Resignation for Good Reason. In the event that, during the Term, the Company terminates Executive’s employment without Cause (other than by reason of death or Disability) or the Executive voluntarily terminates his employment for Good Reason, the Company shall, in full discharge of all of the Company’s obligations to the Executive hereunder or otherwise, provide the Executive with the following payments and benefits: (i) Accrued Rights. The Company shall pay the Executive a lump-sum amount, within thirty (30) days following the Termination Date (or earlier if required by law), equal to the sum of (A) his earned but unpaid Base Salary through the last day of the Executive’s employment (“Termination Date”), (B) any bonus amount earned and vested but not paid for periods ending on or prior to the Termination Date, (C) any accrued and unused PTO per the PTO Policy , (D) any unreimbursed business expenses or other amounts due to the Executive from the Company as of the Termination Date, and (E) all other payments and benefits to which the Executive then may be entitled under the terms of any applicable compensation arrangement or benefit, equity or perquisite plan or program or grant or this Agreement, including but not limited to any applicable insurance benefits (the “Accrued Rights”).

7 (ii) Additional Payments. Subject to Sections 7(d), 7(f) and 7(g) below, the Company shall make additional payments to Executive in the form of continuation of the Executive’s then-current Base Salary (the “Additional Payments”) for a period beginning on the Termination Date and ending on the fifteenth (15) month anniversary of the Termination Date (the “Severance Period”), payable in accordance with the Company’s regular payroll practices, commencing on the Company’s first regular payroll date that occurs on or immediately after the 60th day following the Termination Date; provided, however, the first installment payment of the Additional Payments shall include the cumulative amount of payments that would have been paid to the Executive during the period of time between the Termination Date and the date the Additional Payments commence had such payments commenced immediately following the Termination Date. (iii) COBRA Assistance. If Executive then participates in the Company’s medical and/or dental plans and Executive timely elects to continue and maintain group health plan coverage pursuant to the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), then, subject to Sections 7(f) and 7(g) below, the Company will reimburse Executive monthly for the duration of the Severance Period the cost of such coverage for the Severance Period, which payments will be equal to the amount of the monthly premium for such coverage (the “COBRA Assistance”); provided, however, if the Company is unable to provide the COBRA Assistance without incurring tax penalties or violating applicable nondiscrimination requirements, or the Company terminates the health plans in which Executive is participating and no other health plan is required to provide the COBRA Assistance to Executive, then in lieu of the forgoing benefit, the Company will pay Participant a lump sum cash payment equal to the aggregate cost of the remaining Severance Period for which benefits were to be reimbursed, less any required withholdings within ten (10) business days after Participant’s participation in the health plan terminates. (iv) Bonus Payment. Subject to Sections 7(f) and 7(g) below, the Company shall pay the Executive an amount equal to 125% of 55% of Executive’s Base Salary (at the rate in effect on the Termination Date) payable in equal installments on the Company’s regular payroll dates, commencing on the later of (1) the Company’s first regular payroll date that occurs on or immediately after the 60th day following the Termination Date, and (2) the date that Executive’s Annual Bonus for the calendar year in which the Termination Date occurs would have been paid under Section 3(d) above if Executive had remained employed until the end of such calendar year, and ending on the last payroll date in the Severance Period. (v) Options. The Company shall provide that twenty-five percent (25%) of all outstanding unvested equity awards granted to the Executive shall become fully vested and provide that the time period that the Executive may have to exercise such equity awards shall be extended for a period equal to the shorter of (i) one (1) year following the Termination Date, or (ii) the remaining term of the award. Except as otherwise provided in this Section 7(a)(v), all stock options shall be treated in accordance with the terms of the stock option award and the Company’s Equity Incentive Compensation plan pursuant to which the stock options were granted to the Executive. (vi) Change in Control Benefits. If the Company terminates Executive’s employment without Cause (other than by reason of death or Disability) or the Executive

8 voluntarily terminates his employment for Good Reason during the period commencing on the effective date of a Change in Control and ending on the second anniversary of the effective date of a Change in Control, and subject to Executive complying with his obligations to execute and deliver a Release pursuant to Section 7(e), in addition to the payments and benefits provided under Sections 7(a)(i) and (ii), the Company will (A) extend the Severance Period during which the Additional Payments are made pursuant to Section 7(a)(ii) through the eighteen (18) month anniversary of the Termination Date and (B) the Company shall provide that all outstanding unvested equity awards granted to the Executive shall become fully vested as of the date the Release becomes effective and irrevocable under section 8(e) and provide that the time period that the Executive may have to exercise such equity awards shall be extended for a period equal to the shorter of (i) one (1) year following the Termination Date, and (ii) the remaining term of the award. Except as otherwise provided in this Section 7(a)(v), all stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive.; (C) pay Executive a lump sum amount equal to 150% of Executive’s then annual target bonus on the date the Company pays the first installment of the Additional Payments under Section 7(a)(ii) and (D) increase the COBRA Assistance payable under Section 7(a)(iii) to equal the entire amount of the monthly premium for such coverage, and extend the period of COBRA Assistance through the eighteen (18) month anniversary of the Termination Date. For purposes of this Section, a Change in Control means (v) a transaction or a series of related transactions in which all or substantially all of the assets of the Company are transferred to any “person” or “group” (as such terms are defined in Section 13(d)(3) and 14(d)(2) of the Exchange Act), other than the sale of all or substantially all of the assets of the Company to a person or group who beneficially own, directly or indirectly, at least fifty percent (50%) or more of the combined voting power of the outstanding voting securities of the Company at the time of such sale; (w) any person or group becomes the “beneficial owner” (as defined in Rules 13d-3 and 13d-5 under the Exchange Act), directly or indirectly, of the Company’s outstanding equity representing more than thirty percent (30%) of the total voting power of the Company’s then-outstanding equity; (x) the Company undergoes a merger, reorganization or other consolidation in which the holders of the outstanding equity of the Company immediately prior to such merger, reorganization or consolidation own less than fifty percent (50%) of the surviving entity’s voting power immediately after the transaction; (y) the date a majority of the members of the Company’s incumbent Board is replaced during any twelve (12) month period by members whose appointment or election is not endorsed by a majority to the Company’s incumbent Board before the date of the appointment or election, or (z) the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided further that the Change in Control meets all of the requirements of a “change in the ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(v), a “change in the effective ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(vi), or “a change in the ownership of a substantial portion of the corporation’s assets” within in the meaning of Treasury Regulation §1.409A-3(i)(5)(vii). (b) Resignation without Good Reason, Termination for Cause or upon Death or Disability. (i) In the event that during the Term the Company terminates Executive’s employment for Cause or the Executive voluntarily terminates his employment other than for Good Reason, the Company shall, in full discharge of all of the Company’s obligations to

9 the Executive hereunder or otherwise, pay and/or provide the Executive with any Accrued Rights under Section 7(a)(i) hereof. All stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive. (ii) In the event that during the Term the Executive’s employment is terminated due to the Executive’s death or Disability, the Company shall, in full discharge of all of the Company’s obligations to the Executive (or his estate, if applicable) hereunder or otherwise, (A) pay and/or provide the Executive (or his estate with) with any Accrued Rights under Section 7(a)(i) hereof and (B) subject to Sections 7(f) and 7(g) below, provide the COBRA Assistance under Section 7(a)(iii). All stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive. (c) No Further Rights, Continued Obligations under the Covenants Agreement. The Executive shall have no further rights under this Agreement or otherwise to receive any other compensation or benefits after such termination or resignation of employment under the Company’s severance arrangements or otherwise, except with respect to the payments and benefits specifically provided for under this Section 7. The Executive acknowledges and agrees that, on the expiration of the Term or the earlier termination of his employment for any reason or no reason (whether initiated by the Executive or the Company), the Executive shall continue to be bound by his obligations pursuant to the Covenants Agreement. (d) Payment. Notwithstanding anything in Section 7(a) to the contrary, and subject to Section 19 below, prior to the date the first payment is made under Section 7(a)(ii), Executive may elect to receive a portion of the aggregate amount to which Executive is eligible to receive under Section 7(a)(ii) equal to (i) the product of the compensation limit under Section 401(a)(17), and (ii) two (2), in a lump sum payment on the next payroll period following the Termination Date. For the avoidance of doubt, any payment made pursuant to this Section 7(d) shall be considered a right to receive a series of separate payments (the “First Separation Benefit Plan”), and Executive shall continue to be eligible to receive any amounts pursuant to Section 7(a) in accordance with the terms of Section 7(a)(ii), less any amounts paid under the First Separation Benefit Plan. (e) Release of Claims. Notwithstanding anything contained in this Agreement to the contrary, the Company’s provision of the payments and benefits under Sections 7(a)(ii), 7(a)(iii), 7(a)(iv), 7(a)(v) and 7(a)(vi) hereof shall be contingent in all respects on the Executive (or, if applicable, his estate) executing (and not revoking) a general release of claims against the Company, its affiliates and related parties, in a form reasonably satisfactory to the Company (the “Release”) and the Release becoming effective (and no longer subject to revocation) within sixty (60) days following the Termination Date. (f) Breach of Release or Covenants Agreement. Notwithstanding anything set forth in this Agreement to the contrary, in the event of a breach by the Executive of his obligations under the Covenants Agreement or the Release Agreement and in addition to any other remedies under the Covenants Agreement, the Release Agreement or at law or in equity, the Company shall have no further obligations under Sections 7(a)(ii), 7(a)(iii), 7(a)(iv), or 7(c)(ii) (if and as

10 applicable) and the Executive shall be required, upon demand, to return to the Company any payments previously made by the Company pursuant to Section 7(a)(ii), 7(a)(iii), 7(a)(iv), or 7(c)(ii). (g) Mitigation of Damages. In no event shall the Executive be obliged to seek other employment or take any other action by way of mitigation of the severance benefits payable to the Executive under any of the provisions of this Agreement, nor shall the amount of any severance benefit hereunder be reduced by any compensation earned by the Executive as a result of employment by another employer, except as set forth in this Agreement. Section 8. Covenants Agreement; Corporate Policies. (a) Covenants Agreement. The Executive acknowledges that Executive has executed and remains bound by the Covenants Agreement, which is attached hereto as Exhibit A, the terms of which are incorporated herein by reference, and that the terms of the Covenants Agreement remain in full force and effect and shall survive the expiration of this Agreement or the earlier termination of Executive’s employment hereunder. (b) Corporate Policies. The Executive acknowledges and agrees that during the Term, he will be bound by, and comply with, the Company’s various written corporate policies applicable to other senior executives of the Company, including but not limited to its expense reimbursement policies. Section 9. Withholding Taxes. The Company may directly or indirectly withhold from any payments made under this Agreement all Federal, state, city or other taxes and all other deductions as shall be required pursuant to any law or governmental regulation or ruling or pursuant to any contributory benefit plan maintained by the Company in which the Executive may participate. Section 10. Notices. All notices, requests, demands and other communications required or permitted hereunder shall be given in writing and shall be deemed to have been duly given if delivered or mailed, postage prepaid, by certified or registered mail or by use of an independent third party commercial delivery service for same day or next day delivery and providing a signed receipt as follows: To the Company: Lisata Therapeutics, Inc. 110 Allen Road, 2nd Floor Basking Ridge, NJ 07920 Attention: General Counsel To the Executive: David J. Mazzo, Ph.D. 191 Roundtop Road Bernardsville, NJ 07924-2106

11 or to such other address as either party shall have previously specified in writing to the other. Notice by mail shall be deemed effective on the second business day after its deposit with the United States Postal Service, notice by same day courier service shall be deemed effective on the day of deposit with the delivery service and notice by next day delivery service shall be deemed effective on the day following the deposit with the delivery service. Section 11. No Attachment. Except as required by law, no right to receive payments under this Agreement shall be subject to anticipation, commutation, alienation, sale, assignment, encumbrance, charge, pledge, or hypothecation or to execution, attachment, levy, or similar process or assignment by operation of law, and any attempt, voluntary or involuntary, to effect any such action shall be null, void and of no effect; provided, however, that nothing in this Section 11 shall preclude the assumption of such rights by executors, administrators or other legal representatives of the Executive or his estate and their conveying any rights hereunder to the person or persons entitled thereto. Section 12. Source of Payment. All payments provided for under this Agreement shall be paid in cash from the general funds of the Company. The Company shall not be required to establish a special or separate fund or other segregation of assets to assure such payments, and, if the Company shall make any investments to aid it in meeting its obligations hereunder, the Executive shall have no right, title or interest whatever in or to any such investments except as may otherwise be expressly provided in a separate written instrument relating to such investments. Nothing contained in this Agreement, and no action taken pursuant to its provisions, shall create or be construed to create a trust of any kind, or a fiduciary relationship, between the Company and the Executive or any other person. To the extent that any person acquires a right to receive payments from the Company hereunder, such right, without prejudice to rights which Executives may have, shall be no greater than the right of an unsecured creditor of the Company. Section 13. Binding Agreement; No Assignment. This Agreement shall be binding upon, and shall inure to the benefit of, the Executive and the Company and their respective permitted successors, assigns, heirs, beneficiaries and representatives. This Agreement is personal to the Executive and may not be assigned by him. This Agreement may not be assigned by the Company except in connection with a sale of all or substantially all of its assets or a merger or consolidation of the Company, and the acquiring Company or entity expressly assumes this Agreement. Any attempted assignment in violation of this Section 13 shall be null and void. Section 14. Governing Law; Consent to Jurisdiction. The validity, interpretation, performance, and enforcement of this Agreement shall be governed by the laws of the State of New Jersey. In addition, the Executive and the Company irrevocably submit to the exclusive jurisdiction of the courts of the State of New Jersey and the United States District Court sitting in New Jersey for the purpose of any suit, action, proceeding or judgment relating to or arising out of this Agreement and the transactions contemplated hereby. Service of process in connection with any such suit, action or proceeding may be served on the Executive or the Company anywhere in the world by the same methods as are specified for the giving of notices under this Agreement. The Executive and the Company irrevocably consent to the jurisdiction of any such court in any such suit, action or proceeding and to the laying of venue in such court. In any such action or proceeding, the court shall have the authority to award reasonable costs, expenses, and attorneys’ fees to the party that substantially prevails.

12 Section 15. Entire Agreement; Amendments. This Agreement (including Exhibit A) embodies the entire agreement between Executive and the Company with respect to the subject matter hereof and may only be amended or otherwise modified by a writing executed by all of the parties hereto. Section 16. Counterparts. This Agreement may be executed in any number of counterparts, each of which when executed shall be deemed to be an original and all of which together shall be deemed to be one and the same instrument. Section 17. Severability; Blue-Penciling. The provisions, sections and paragraphs, and the specific terms set forth therein, of this Agreement (including Exhibit A) are severable, except as specifically provided to the contrary herein. If any provision, section or paragraph, or specific term contained therein, of this Agreement or the application thereof is determined by a court to be illegal, invalid or unenforceable, that provision, section, paragraph or term shall not be a part of this Agreement, and the legality, validity and enforceability of remaining provisions, sections and paragraphs, and all other terms therein, of this Agreement shall not be affected thereby. The Executive acknowledges and agrees that as to himself, the restrictive covenants contained in the Covenants Agreement (the “Restrictive Covenants”) are reasonable and valid in geographical and temporal scope and in all other respects. If any court determines that any of such Restrictive Covenants, or any part thereof, is invalid or unenforceable, the remainder of the Restrictive Covenants shall not thereby be affected and shall be given full effect, without regard to the invalid portions. It is the desire and intent of the parties that the Restrictive Covenants will be enforced to the fullest extent permissible under the laws and public policies applied in each jurisdiction in which enforcement is sought. Accordingly, if any Restrictive Covenant shall be adjudicated to be invalid or unenforceable, such Restrictive Covenant shall be deemed amended to the extent necessary in order that such provision be valid and enforceable, such amendment to apply only with respect to the operation of such Restrictive Covenant in the particular jurisdiction in which such adjudication is made. Section 18. Prior Agreements. This Agreement supersedes all prior agreements and understandings (including verbal agreements) between Executive and the Company regarding the terms and conditions of Executive’s employment with the Company. Section 19. 409A Compliance. (a) It is intended that each installment of the payments and benefits provided under Section 7 of this Agreement shall be treated as a separate “payment” for purposes of Section 409A of the United States Internal Revenue Code of 1986, as amended (the “Code”). (b) Notwithstanding anything contained herein to the contrary, the Executive shall not be considered to have terminated employment with the Company for purposes of Section 6 and 7 of this Agreement unless he would be considered to have incurred a “separation from service” from the Company within the meaning of Treasury Regulation §1.409A-1(h)(1)(ii). (c) The severance payments set forth in Section 7(a)(i) of this Agreement are intended to comply with the involuntary separation pay plan exception to Section 409A set forth in Treas. Reg. §1.409A-1(b)(9)(iii) to the extent that the payments due under Section 7(a) are in

13 the aggregate less than the lesser of two times the lesser of (i) the sum of the Executive’s annualized compensation based upon the annual rate of pay for services provided to the Company for the taxable year of the Company preceding the taxable year in which the Termination Date occurs (adjusted for any increase during that year that was expected to continue indefinitely if Executive had not separated from service) and (ii) the applicable compensation limit under Section 401(a)(17) of the Code for the year in which the Termination Date occurs shall be in accordance with as an involuntary separation plan. (d) The payment set forth in Section 7(a)(iv) of this Agreement is intended to comply with the short-term deferral exception to Section 409A of the Code set forth in Treas. Reg. §1.409A-1(b)(4). (e) All references in this Agreement to Executive’s termination of employment shall mean Executive’s “separation from service” within the meaning of Section 409A of the Code and Treas. Reg. §1.409A-1(h). (f) Executive acknowledges and agrees that notwithstanding any provision of this Agreement, the Company and its affiliates are not providing Executive with any tax advice with respect to Section 409A of the Code or otherwise and are not making any guarantees or other assurances of any kind to Executive with respect to the tax consequences or treatment of any amounts paid or payable to Executive under this Agreement. (g) Notwithstanding anything to the contrary contained herein, if necessary to comply with the restriction in Section 409A(a)(2)(B) of the Code concerning payments to “specified Executives,” any payment on account of the Executive’s separation from service that would otherwise be due hereunder within six (6) months after such separation shall nonetheless be delayed until the first business day of the seventh month following the Executive’s date of termination and the first such payment shall include the cumulative amount of any payments that would have been paid prior to such date if not for such restriction, together with interest on such cumulative amount during the period of such restriction at a rate, per annum, equal to the applicable federal short-term rate (compounded monthly) in effect under Section 1274(d) of the Code on the date of termination. If the Executive dies during the six-month postponement period prior to the payment, the amount of the payment deferred on account of Section 409A of the Code shall be paid to the personal representative of the Executive’s estate within thirty (30) days after the date of the Executive’s death. For purposes of Section 7 hereof, the Executive shall be a “specified Executive” for the 12-month period beginning on the first day of the fourth month following each “Identification Date” if he is a “key Executive” (as defined in Section 416(i) of the Code without regard to Section 416(i)(5) thereof) of the Company at any time during the 12-month period ending on the “Identification Date.” For purposes of the foregoing, the Identification Date shall be December 31. (h) To the extent that any provision in this Agreement is ambiguous as to its compliance with Section 409A, the provision shall be read in such a manner so that no payments due under this Agreement shall be subject to an “additional tax” as defined in Section 409A(a)(1)(B) of the Code.

14 (i) All reimbursements provided under this Agreement shall be made or provided in accordance with the requirements of Section 409A, including, where applicable, the requirement that (i) any reimbursement is for expenses incurred during the Executive’s lifetime (or during a shorter period of time specified in this Agreement), (ii) the amount of expenses eligible for reimbursement during a calendar year may not affect the expenses eligible for reimbursement in any other calendar year, (iii) the reimbursement of an eligible expense will be made on or before the last day of the calendar year following the year in which the expense is incurred, and (iv) the right to reimbursement is not subject to liquidation or exchange for another benefit. (j) In no event whatsoever shall the Company be liable for any additional tax, interest or penalty that may be imposed on Executive by Section 409A or damages for failing to comply with Section 409A. Section 20. Section 280G Limitation. If any payment(s) or benefit(s) the Executive would receive pursuant to this Agreement and/or pursuant to any other agreement or arrangement would (a) constitute a “parachute payment” within the meaning of Section 280G of the Code, (b) but for this Section 20, be subject to the excise tax imposed by Section 4999 of the Code (the “Excise Tax”), and (c) if the net-after tax amount (taking into account all applicable taxes payable by the Executive, including any Excise Tax) that the Executive would receive with respect to such payments or benefits does not exceed the Reduced Amount, then such payment(s) or benefit(s) (collectively, “Payments”) shall be reduced to the Reduced Amount. The “Reduced Amount” shall be the largest portion of the Payments that can be paid or provided without causing any portion of the Payments being subject to the Excise Tax. If a reduction in payments or benefits constituting “parachute payments” is necessary so that the Payments equal the Reduced Amount, reduction shall occur in the following order: (i) first, any base salary severance payments; (ii) second, any other cash payments due under any other agreement between the Company and the Executive; (iii) third, cancellation of the acceleration of vesting of any stock options; (iv) fourth, cancellation of the acceleration of vesting of any restricted stock and restricted stock units; and (v) lastly, other non-cash forms of benefits. Calculations of the foregoing will be performed at the expense of the Company by an accounting firm selected by the Company. The determinations of such accounting firm shall be final, binding and conclusive upon the Company and the Executive. [Signatures follow on next page]

15 IN WITNESS WHEREOF, the Company has caused this Agreement to be executed by their respective duly authorized officers and the Executive has signed this Agreement, all as of the first date above written but effective as of the Effective Date. LISATA THERAPEUTICS, INC. By: /s/ Steven M. Klosk Name: Steven M. Klosk Title: Chairman of the Compensation Committee Lisata Therapeutics, Inc. Board of Directors /s/ David J. Mazzo David J. Mazzo, Ph.D.

1 Exhibit 10.2 AMENDED AND RESTATED EMPLOYMENT AGREEMENT This AMENDED AND RESTATED EMPLOYMENT AGREEMENT (this “Agreement”), dated as of June 10, 2025, and effective on June 10, 2025 (the “Effective Date”) is by and between Lisata Therapeutics, Inc. (the “Company”) and Kristen K. Buck, M.D. (the “Executive”) of 727 Creek Road, Warminster, PA 18974. WITNESSETH: WHEREAS, the Company desires to continue to employ the Executive as its Executive Vice President, R&D and Chief Medical Officer and the Executive desires to be so employed by the Company; and WHEREAS, the Company and the Executive each believe it is in their respective best interests to enter into this Agreement setting forth the mutual understandings and agreements reached between the Company and the Executive with respect to the Executive’s employment with the Company and certain restrictions on the Executive’s conduct benefiting the Company during such time and thereafter, all as set forth herein. NOW, THEREFORE, in consideration of the promises and the mutual covenants and agreements contained herein and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto, intending to be legally bound hereby, agree as follows: Section 1. Condition to Enforceability. The Executive hereby represents and warrants to the Company that she has the legal capacity to execute and perform this Agreement, and that its execution and performance by her will not violate the terms of any existing agreement or understanding to which the Executive is a party; and the Company hereby represents and warrants to the Executive that the person executing this Agreement on its behalf has the authority to do so and to bind the Company. Section 2. Employment. The Company agrees to employ the Executive, and the Executive agrees to be employed by the Company on a full-time basis, for the period commencing on the Effective Date (the date on which the Executive actually begins her employment, the “Start Date”) and, subject to earlier termination pursuant to Section 7 below, continuing until the three year anniversary of the Start Date (the “Initial Term”). Unless Executive is given written notice by the Company (acting through the Company’s Board of Directors (the “Board”)) no later than ninety (90) days prior to the expiration of the Initial Term, Executive’s employment hereunder shall be deemed extended for an additional period of one (1) year, subject to earlier termination pursuant to Section 7 below (each, a “Renewal Term”), in each such case, commencing upon the expiration of the Initial Term or the then-current Renewal Term, as the case may be. As used in this Agreement, the “Term” shall refer to the period beginning on the Start Date and ending on the effective date of the termination of this Agreement and the Executive’s employment hereunder (the “Termination Date”) in accordance with this Section or Section 7 below.

2 Section 3. Position and Duties. During the Term, the Executive shall be employed as the Company’s Executive Vice President, R&D and Chief Medical Officer and shall perform duties consistent with such position, including but not limited to direct responsibility for all day-to-day operations of the Research and Development Organization and such other related duties as the President and Chief Executive Officer shall reasonably request. During the Term, and except for PTO in accordance with Section 6(a) below, the Executive shall devote her full business time, attention, skill and efforts to the business and affairs of the Company, its subsidiaries and other affiliates and shall comply with the Company’s codes of conduct, policies and procedures as applicable at any given time. The Executive will have the opportunity to serve on no more than two external private or public company boards with permission of the Board, which shall not be unreasonably withheld, provided that the company does not compete with the business of the Company and the Executive’s outside services do not materially interfere, as determined by the Board in good faith, with the performance of the Executive’s duties and responsibilities hereunder. Section 4. Compensation. For all services rendered by the Executive in any capacity required hereunder during the Term, the Executive shall be compensated as follows: (a) The Company shall pay the Executive a base salary (the “Base Salary”) at the annualized rate of five hundred ninety-nine thousand three hundred forty-two dollars ($599,342) which shall be subject to customary withholdings and authorized deductions and be payable in equal installments in accordance with the Company’s customary payroll practices in place from time to time. The Executive’s Base Salary shall be subject to review by the Board and/or the Compensation Committee thereof (the “Compensation Committee”) at least annually and may be increased, but not decreased, from time to time by the Board. As used in this Agreement, the term “Base Salary” shall refer to base salary as may be adjusted from time to time. (b) The Executive shall be entitled to participate in all compensation and employee benefit plans or programs and to receive all other benefits and perquisites that are approved by the Board and are generally made available by the Company to other senior executives of the Company and to the extent permissible under the general terms and provisions of such plans or programs and in accordance with the provisions thereof. Notwithstanding any of the foregoing, nothing in this Agreement shall require the Company or any subsidiary or affiliate thereof to establish, maintain or continue any particular plan or program nor preclude the amendment, rescission or termination of any such plan or program that may be established from time to time. (c) The Executive is eligible to receive an annual equity award, the size and value of which to be determined by the Board (or the Compensation Committee) in its discretion. Any equity award will be granted in accordance with the terms of the applicable equity plan or arrangement that may be in effect from time to time. (d) The Executive shall be eligible to receive an annual cash bonus for each calendar year ending during the Term (“Annual Bonus”). The Executive’s target Annual Bonus will equal 50% of her Base Salary (the “Target Bonus”). The amount of the Annual Bonus awarded to you will be determined by the Board and/or the Compensation Committee thereof, in its sole discretion, based upon the level of achievement of the Company’s corporate goals for the

3 corresponding calendar year along with the performance of the Executive in relation to her personal performance objectives. Accordingly, the actual Annual Bonus may be less than or greater than the Target Bonus. Each Annual Bonus for a calendar year, to the extent earned, will be paid in a lump sum early in the subsequent calendar year, but no later than March 15. In order for the Executive to receive an Annual Bonus, the Executive must be actively employed by the Company on December 31 of the calendar year for which the Annual Bonus is applicable. Section 5. Business Expenses. The Company shall pay or reimburse the Executive for all reasonable travel and other reasonable expenses incurred by the Executive in connection with the performance of her duties and obligations under this Agreement, subject to the Executive’s presentation of appropriate vouchers or receipts in accordance with such policies and approval procedures as the Company may from time to time establish for employees (including but not limited to prior approval of extraordinary expenses) and to preserve any deductions for Federal income taxation purposes to which the Company may be entitled. Section 6. Benefits; Perquisites; Expense Reimbursement. In addition to those payments and benefits set forth above or elsewhere herein, the Executive shall be entitled to the following other benefits and payments: (a) Vacation. Executive shall be entitled to twenty-nine (29) days of paid time off (“PTO”) per calendar year (prorated in the event of a service year which is shorter than a calendar year) under the Company’s then in effect PTO Policy, in addition to Company-observed holidays. Any PTO time not used during a calendar year shall be treated in accordance with the Company’ s policies relating to unused PTO time. (b) D&O Insurance. The Executive shall be covered by the Directors and Officers Liability Insurance policy that generally covers the directors and officers of the Company on the same terms and conditions provided to the Company’s other executive offers, provided by the Company at its expense. (c) Indemnification. The Executive shall be entitled to the benefit of the indemnification provisions contained in the Company’s By-laws or Certificate of Incorporation as they may be amended from time to time, to the extent permitted by applicable law, at the time of the assertion of any liability against the Executive. For clarity, the Company will not indemnify the Executive in connection with any liability arising out of or related to her employment relationship with any predecessor employer. Section 7. Termination of Employment. (a) Events of Termination. The Executive’s employment hereunder may be terminated upon the occurrence of any of the following events: (i) Termination for Cause. The Company (acting through the Board) may terminate the Executive’s employment hereunder for Cause at any time. For purposes of this Agreement, “Cause” shall mean that, as determined by the Board, the Executive has:

4 (A) committed gross negligence in connection with her duties as set forth herein or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (B) committed fraud in connection with her duties as set forth herein or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (C) engaged in personal dishonesty, willful misconduct, willful violation of any law, or breach of fiduciary duty, in each instance, with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (D) been indicted for, or has been found by a court of competent jurisdiction to have committed or plead guilty to (1) a felony (or state law equivalent) or (2) any other serious crime involving moral turpitude or that has (or is reasonably likely to have) a material adverse effect either on (x) the Executive’ s ability to perform her duties under the Agreement or (y) the reputation and goodwill of the Company, regardless of whether or not such other crime is related or unrelated to the business of the Company, its subsidiaries or other affiliates; (E) shown chronic use of alcohol, drugs or other similar substances that materially affects the Executive’s work performance; (F) breached her obligations under (1) this Agreement, (2) the Confidentiality, Non-Compete and Inventions Assignment Agreement attached hereto as Exhibit A (the “Covenants Agreement”), or (3) any other agreement executed by the Executive for the benefit of the Company, its subsidiaries and/or other affiliates, provided, that, if such breach described in this clause (F) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to cure such breach; (G) failed to materially perform the Executive’s duties or to follow the lawful directives of the Board; provided, that, if such failure described in this clause (G) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to cure such failure; or (H) materially violated the Company’s written code of conduct or other written or established policies and/or procedures in place from time to time; provided, that, if such violation described in this clause (H) is susceptible to cure (as determined in the reasonable discretion of the Board), the Executive shall have thirty (30) days after notice from the Board to cure such violation. Any notice to the Executive under this Section 7(a)(i) shall be in writing and shall specify in reasonable detail the Executive’s acts or omissions that the Company alleges constitute “Cause.” (ii) Termination without Cause. The Company (acting through the Board) may terminate the Executive’s employment hereunder without Cause (other than by reason of death or Disability) at any time upon notice to Executive.

5 (iii) Resignation for Good Reason. The Executive may voluntarily terminate her employment hereunder for Good Reason (as defined below) upon written notice to the Company in accordance with the definition thereof. For purposes of this Agreement, “Good Reason” shall mean the occurrence of any of the following events: (A) material breach by the Company of its obligations under this Agreement; (B) the Executive’s base salary, position, duties, responsibilities, or authority have been materially reduced or the Executive has repeatedly been assigned duties that are materially inconsistent with her duties set forth herein, in each case, without the Executive’s consent or (C) the relocation of the Executive’s principal place of employment, without the Executive’s consent, in a manner that lengthens her one-way commute distance by fifty (50) or more miles. “Good Reason” shall not be deemed to exist, however, unless (1) the Executive shall have given written notice to the Company specifying in reasonable detail the Company’s acts or omissions that the Executive alleges constitute “Good Reason” within sixty (60) days after the first occurrence of such circumstances and the Company shall have failed to cure any such act or omission within sixty (60) days of receipt of such written notice, and (2) the Executive actually terminates employment within one hundred eighty (180) days following the initial occurrence of any of the foregoing conditions that she considers to be “Good Reason.” If the Executive fails to provide this notice and cure period prior to her resignation or resigns more than one hundred eighty (180) days after the initial existence of the condition, her resignation will not be deemed to be for “Good Reason.” (iv) Resignation without Good Reason. The Executive may voluntarily terminate her employment hereunder for any reason at any time for any reason that does not constitute Good Reason, upon thirty (30) days’ prior written notice to the Company, provided, however, the Company reserves the right, upon written notice to the Executive, to accept the Executive’s notice of resignation and to accelerate such notice and make the Executive’s resignation effective immediately, or on such other date prior to the Executive’s intended last day of work as the Company deems appropriate. It is understood and agreed that the Company’s election to accelerate Executive’s notice of resignation shall not be deemed a termination by the Company without Cause for purposes of Section 7(a)(ii) of this Agreement, Section 8(a) of this Agreement or otherwise, or constitute Good Reason for purposes of Section 7(a)(iii) of this Agreement, Section 8(a) of this Agreement or otherwise. (v) Disability. The Executive’s employment hereunder shall terminate upon her Disability. For purposes of this Agreement, “Disability” shall mean that the Executive has been unable to perform her duties to the Company on account of physical or mental illness or incapacity for a period of ninety (90) consecutive calendar days or one hundred twenty (120) calendar days (whether or not consecutive) during any 365-day period, as a result of a condition that is treated as a total or permanent disability under the long-term disability insurance policy of the Company that covers the Executive. (vi) Death. The Executive’s employment hereunder shall automatically terminate upon her death. (b) Resignation from Directorships, Officerships and Committees. The termination of the Executive’s employment for any reason shall constitute the Executive’s resignation from (i) any director, officer, employee or committee position the Executive has with the Company or any of its affiliates and (ii) all fiduciary positions the Executive holds with respect

6 to any employee benefit plans or trusts established by the Company. The Executive agrees that this Agreement shall serve as written notice of resignation in this circumstance; provided, however, the Executive agrees to take any additional actions that are deemed reasonably necessary by the Company to effectuate or evidence such resignations. (c) Interpretation. For purposes of this Section 7, the Board will determine in its reasonable discretion whether a termination of Executive’s employment constitutes a termination with Cause under Section 7(a)(i), a termination without Cause under Section 7(a)(ii), a resignation with Good Reason under Section 7(a)(iii), or a resignation without Good Reason under Section 7(a)(iv). If the Company consummates a Change in Control, the members of the Board as constituted immediately prior to the closing of a Change in Control (the “Incumbent Board”) shall retain authority to make the forgoing determinations concerning the termination of Executive’s employment until the second (2nd) anniversary of the closing date of the Change in Control regardless of whether the Company’s Board is reconstituted in connection with the Change in Control. Following a Change in Control, the Incumbent Board may delegate its authority under this Section 7(c) to a qualified independent third party, such as a retired judge selected by the head of the American Arbitration Association in Manhattan, an independent compensation consultant or a law firm. Section 8. Compensation upon Termination of Employment. All defined terms used in this Section but not defined in this Section or elsewhere in this Agreement shall have the meanings ascribed to such terms in the Covenants Agreement: (a) Termination without Cause; Resignation for Good Reason. In the event that, during the Term, the Company terminates Executive’s employment without Cause (other than by reason of death or Disability) or the Executive voluntarily terminates her employment for Good Reason, the Company shall, in full discharge of all of the Company’s obligations to the Executive hereunder or otherwise. provide the Executive with the following payments and benefits: (i) Accrued Rights. The Company shall pay the Executive a lump-sum amount, within thirty (30) days following the Termination Date (or earlier if required by law), equal to the sum of (A) her earned but unpaid Base Salary through the last day of the Executive’s employment (“Termination Date”), (B) any bonus amount earned and vested but not paid for periods ending on or prior to the Termination Date, (C) any accrued and unused PTO per the PTO Policy, (D) any unreimbursed business expenses or other amounts due to the Executive from the Company as of the Termination Date, and (E) all other payments and benefits to which the Executive then may be entitled under the terms of any applicable compensation arrangement or benefit, equity or perquisite plan or program or grant or this Agreement, including but not limited to any applicable insurance benefits (the “Accrued Rights”). (ii) Additional Payments. Subject to Sections 8(d), 8(f) and 8(g) below, the Company shall make additional payments to Executive in the form of continuation of the Executive’s then-current Base Salary (the “Additional Payments”) for a period beginning on the Termination Date and ending on the twelve (12) month anniversary of the Termination Date (the “Severance Period”), payable in accordance with the Company’s regular payroll practices, commencing on the Company’s first regular payroll date that occurs on or immediately after the 60th day following the Termination Date; provided, however, the first installment payment of the

7 Additional Payments shall include the cumulative amount of payments that would have been paid to the Executive during the period of time between the Termination Date and the date the Additional Payments commence had such payments commenced immediately following the Termination Date. (iii) COBRA Assistance. If Executive then participates in the Company’s medical and/or dental plans and Executive timely elects to continue and maintain group health plan coverage pursuant to the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), then, subject to Sections 8(f) and 8(g) below, the Company will reimburse Executive monthly for the duration of the Severance Period the cost of such coverage for the Severance Period, which payments will be equal to the amount of the monthly premium for such coverage (the “COBRA Assistance”); provided, however, if the Company is unable to provide the COBRA Assistance without incurring tax penalties or violating applicable nondiscrimination requirements or the Company terminates the health plans in which Executive is participating and no other health plan is required to provide the COBRA Assistance to Executive, then in lieu of the forgoing benefit, the Company will pay Participant a lump sum cash payment equal to the aggregate cost of the remaining Severance Period for which benefits were to be reimbursed, less any required withholdings within ten (10) business days after Participant’s participation in the health plan terminates. (iv) Bonus Payment. Subject to Sections 8(f) and 8(g) below, the Company shall pay the Executive an amount equal to the Target Bonus in effect for the year in which Executive’s termination occurs, payable in equal installments on the Company’s regular payroll dates, commencing on the later of (1) the Company’s first regular payroll date that occurs on or immediately after the 60th day following the Termination Date, and (2) the date that Executive’s Annual Bonus for the calendar year in which the Termination Date occurs would have been paid under Section 4(d) above if Executive had remained employed until the end of such calendar year, and ending on the last payroll date in the Severance Period. (v) Options. The Company shall provide that the time period that the Executive may have to exercise any fully vested option equity awards shall be extended for a period equal to the shorter of (i) one (1) year following the Termination Date, or (ii) the remaining term of the award. Except as otherwise provided in this Section 8(a)(v), all stock options shall be treated in accordance with the terms of the stock option award and the Company’s Equity Incentive Compensation plan pursuant to which the stock options were granted to the Executive. (vi) Change in Control Benefits. If the Company terminates Executive’s employment without Cause (other than by reason of death or Disability) or the Executive voluntarily terminates her employment for Good Reason during the period commencing on the effective date of a Change in Control and ending on the second anniversary of the effective date of a Change in Control, and subject to Executive complying with her obligations to execute and deliver a Release pursuant to Section 8(e), in addition to the payments and benefits provided under Sections 8(a)(i) and (ii), the Company will: (A) extend the Severance Period during which the Additional Payments are made pursuant to Section 8(a)(ii) through the fifteen (15) month anniversary of the Termination Date, and (B) the Company shall provide that all outstanding unvested time-based equity awards granted to the Executive shall become fully vested as of the date the Release becomes effective and irrevocable under Section 8(e) and provide that the time

8 period that the Executive may have to exercise such option equity awards shall be extended for a period equal to the shorter of (i) one (1) year following the Termination Date, and (ii) the remaining term of the award. Except as otherwise provided in this Section 8(a)(vi), all stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive; (C) pay Executive a lump sum amount equal to 125% of Executive’s then Target Bonus on the date the Company pays the first installment of the Additional Payments under Section 8(a)(ii), and (D) increase the COBRA Assistance payable under Section 8(a)(iii) to equal the entire amount of the monthly premium for such coverage, and extend the period of COBRA Assistance through the fifteen (15) month anniversary of the Termination Date. For purposes of this Section, a “Change in Control” means (v) a transaction or a series of related transactions in which all or substantially all of the assets of the Company are transferred to any “person” or “group” (as such terms are defined in Section 13(d)(3) and 14(d)(2) of the Exchange Act), other than the sale of all or substantially all of the assets of the Company to a person or group who beneficially own, directly or indirectly, at least fifty percent (50%) or more of the combined voting power of the outstanding voting securities of the Company at the time of such sale; (w) any person or group becomes the “beneficial owner” (as defined in Rules 13d-3 and 13d-5 under the Exchange Act), directly or indirectly, of the Company’s outstanding equity representing more than thirty percent (30%) of the total voting power of the Company’s then-outstanding equity; (x) the Company undergoes a merger, reorganization or other consolidation in which the holders of the outstanding equity of the Company immediately prior to such merger, reorganization or consolidation own less than fifty percent (50%) of the surviving entity’s voting power immediately after the transaction; (y) the date a majority of the members of the Company’s incumbent Board is replaced during any twelve (12) month period by members whose appointment or election is not endorsed by a majority to the Company’s incumbent Board before the date of the appointment or election, or (z) the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided further that the Change in Control meets all of the requirements of a “change in the ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(v), a “change in the effective ownership of a corporation” within the meaning of Treasury Regulation §1.409A- 3(i)(5)(vi), or “a change in the ownership of a substantial portion of the corporation’s assets” within in the meaning of Treasury Regulation §1.409A-3(i)(5)(vii). (b) Resignation without Good Reason, Termination for Cause or upon Death or Disability (i) In the event that during the Term the Company terminates Executive’s employment for Cause or the Executive voluntarily terminates her employment other than for Good Reason, the Company shall, in full discharge of all of the Company’s obligations to the Executive hereunder or otherwise, pay and/or provide the Executive with any Accrued Rights under Section 8(a)(i) hereof. All stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive. (ii) In the event that during the Term the Executive’s employment is terminated due to the Executive’s death or Disability, the Company shall, in full discharge of all of the Company’s obligations to the Executive (or her estate, if applicable) hereunder or otherwise, (A) pay and/or provide the Executive (or her estate with) with any Accrued Rights under

9 Section 8(a)(i) hereof, and (B) subject to Sections 8(f) and 8(g) below, provide the COBRA Assistance under Section 8(a)(iii). All stock options shall be treated in accordance with the terms of the stock option award and the Company’s equity incentive plan pursuant to which the stock options were granted to the Executive. (c) No Further Rights, Continued Obligations under the Covenants Agreement. The Executive shall have no further rights under this Agreement or otherwise to receive any other compensation or benefits after such termination or resignation of employment under the Company’s severance arrangements or otherwise, except with respect to the payments and benefits specifically provided for under this Section 8. The Executive acknowledges and agrees that, on the expiration of the Term or the earlier termination of her employment for any reason or no reason (whether initiated by the Executive or the Company), the Executive shall continue to be bound by her obligations pursuant to the Covenants Agreement. (d) Payment. Notwithstanding anything in Section 8(a) to the contrary, and subject to Section 20 below, prior to the date the first payment is made under Section 8(a)(ii), Executive may elect to receive a portion of the aggregate amount to which Executive is eligible to receive under Section 8(a)(ii) equal to (i) the product of the compensation limit under Section 401(a)(17), and (ii) two (2), in a lump sum payment on the next payroll period following the Termination Date. For the avoidance of doubt, any payment made pursuant to this Section 8(d) shall be considered a right to receive a series of separate payments (the “First Separation Benefit Plan”), and Executive shall continue to be eligible to receive any amounts pursuant to Section 7(a) in accordance with the terms of Section 8(a)(ii), less any amounts paid under the First Separation Benefit Plan. (e) Release of Claims. Notwithstanding anything contained in this Agreement to the contrary, the Company’s provision of the payments and benefits under Sections 8(a)(ii), 8(a)(iii), 8(a)(iv), 8(a)(v), and 8(a)(vi) hereof shall be contingent in all respects on the Executive (or, if applicable, her estate) executing (and not revoking) a general release of claims against the Company, its affiliates and related parties, in a form reasonably satisfactory to the Company (the “Release”) and the Release becoming effective (and no longer subject to revocation) within sixty (60) days following the Termination Date. (f) Breach of Release or Covenants Agreement. Notwithstanding anything set forth in this Agreement to the contrary, in the event of a material breach by the Executive of her obligations under the Covenants Agreement or the Release Agreement and in addition to any other remedies under the Covenants Agreement, the Release Agreement or at law or in equity, the Company shall have no further obligations under Sections 8(a)(ii), 8(a)(iii), 8(a)(iv), or 8(c)(ii) (if and as applicable) and the Executive shall be required, upon demand, to return to the Company any payments previously made by the Company pursuant to Section 8(a)(ii), 8(a)(iii), 8(a)(iv), or 8(c)(ii). (g) Mitigation of Damages. In no event shall the Executive be obliged to seek other employment or take any other action by way of mitigation of the severance benefits payable to the Executive under any of the provisions of this Agreement, nor shall the amount of any severance benefit hereunder be reduced by any compensation earned by the Executive as a result of employment by another employer, except as set forth in this Agreement.

10 Section 9. Covenants Agreement; Corporate Policies. (a) Covenants Agreement. The Executive acknowledges that Executive has executed and remains bound by the Covenants Agreement, which is attached hereto as Exhibit A, the terms of which are incorporated herein by reference, and that the terms of the Covenants Agreement remain in full force and effect and shall survive the expiration of this Agreement or the earlier termination of Executive’s employment hereunder. (b) Corporate Policies. The Executive acknowledges and agrees that during the Term, she will be bound by, and comply with, the Company’s various written corporate policies applicable to other senior executives of the Company, including but not limited to its expense reimbursement policies. Section 10. Withholding Taxes. The Company may directly or indirectly withhold from any payments made under this Agreement all Federal, state, city or other taxes and all other deductions as shall be required pursuant to any law or governmental regulation or ruling or pursuant to any contributory benefit plan maintained by the Company in which the Executive may participate. Section 11. Notices. All notices, requests, demands and other communications required or permitted hereunder shall be given in writing and shall be deemed to have been duly given if delivered or mailed, postage prepaid, by certified or registered mail or by use of an independent third party commercial delivery service for same day or next day delivery and providing a signed receipt as follows: To the Company: Lisata Therapeutics, Inc. 110 Allen Road, 2nd Floor Basking Ridge, NJ 07920 Attention: General Counsel To the Executive: Kristen K. Buck, M.D. 727 Creek Road Warminster, PA 18974 or to such other address as either party shall have previously specified in writing to the other. Notice by mail shall be deemed effective on the second business day after its deposit with the United States Postal Service, notice by same day courier service shall be deemed effective on the day of deposit with the delivery service and notice by next day delivery service shall be deemed effective on the day following the deposit with the delivery service. Section 12. No Attachment. Except as required by law, no right to receive payments under this Agreement shall be subject to anticipation, commutation, alienation, sale, assignment, encumbrance, charge, pledge, or hypothecation or to execution, attachment, levy, or similar process or assignment by operation of law, and any attempt, voluntary or involuntary, to effect any

11 such action shall be null, void and of no effect; provided, however, that nothing in this Section shall preclude the assumption of such rights by executors, administrators or other legal representatives of the Executive or her estate and their conveying any rights hereunder to the person or persons entitled thereto. Section 13. Source of Payment. All payments provided for under this Agreement shall be paid in cash from the general funds of the Company. The Company shall not be required to establish a special or separate fund or other segregation of assets to assure such payments, and, if the Company shall make any investments to aid it in meeting its obligations hereunder, the Executive shall have no right, title or interest whatever in or to any such investments except as may otherwise be expressly provided in a separate written instrument relating to such investments. Nothing contained in this Agreement, and no action taken pursuant to its provisions, shall create or be construed to create a trust of any kind, or a fiduciary relationship, between the Company and the Executive or any other person. To the extent that any person acquires a right to receive payments from the Company hereunder, such right, without prejudice to rights which Executives may have, shall be no greater than the right of an unsecured creditor of the Company. Section 14. Binding Agreement; No Assignment. This Agreement shall be binding upon, and shall inure to the benefit of, the Executive and the Company and their respective permitted successors, assigns, heirs, beneficiaries and representatives. This Agreement is personal to the Executive and may not be assigned by her. This Agreement may not be assigned by the Company except in connection with a sale of all or substantially all of its assets or a merger or consolidation of the Company, and the acquiring Company or entity expressly assumes this Agreement. Any attempted assignment in violation of this Section shall be null and void. Section 15. Governing Law; Consent to Jurisdiction. The validity, interpretation, performance, and enforcement of this Agreement shall be governed by the laws of the State of New Jersey. In addition, the Executive and the Company irrevocably submit to the exclusive jurisdiction of the courts of the State of New Jersey and the United States District Court sitting in New Jersey for the purpose of any suit, action, proceeding or judgment relating to or arising out of this Agreement and the transactions contemplated hereby. Service of process in connection with any such suit, action or proceeding may be served on the Executive or the Company anywhere in the world by the same methods as are specified for the giving of notices under this Agreement. The Executive and the Company irrevocably consent to the jurisdiction of any such court in any such suit, action or proceeding and to the laying of venue in such court. In any such action or proceeding, the court shall have the authority to award reasonable costs, expenses, and attorneys’ fees to the party that substantially prevails. Section 16. Entire Agreement; Amendments. This Agreement (including Exhibit A) embodies the entire agreement between Executive and the Company with respect to the subject matter hereof and may only be amended or otherwise modified by a writing executed by all of the parties hereto. Section 17. Counterparts. This Agreement may be executed in any number of counterparts, each of which when executed shall be deemed to be an original and all of which together shall be deemed to be one and the same instrument.

12 Section 18. Severability; Blue-Penciling. The provisions, sections and paragraphs, and the specific terms set forth therein, of this Agreement (including Exhibit A) are severable, except as specifically provided to the contrary herein. If any provision, section or paragraph, or specific term contained therein, of this Agreement or the application thereof is determined by a court to be illegal, invalid or unenforceable, that provision, section, paragraph or term shall not be a part of this Agreement, and the legality, validity and enforceability of remaining provisions, sections and paragraphs, and all other terms therein, of this Agreement shall not be affected thereby. The Executive acknowledges and agrees that as to herself, the restrictive covenants contained in the Covenants Agreement (the “Restrictive Covenants”) are reasonable and valid in geographical and temporal scope and in all other respects. If any court determines that any of such Restrictive Covenants, or any part thereof, is invalid or unenforceable, the remainder of the Restrictive Covenants shall not thereby be affected and shall be given full effect, without regard to the invalid portions. It is the desire and intent of the parties that the Restrictive Covenants will be enforced to the fullest extent permissible under the laws and public policies applied in each jurisdiction in which enforcement is sought. Accordingly, if any Restrictive Covenant shall be adjudicated to be invalid or unenforceable, such Restrictive Covenant shall be deemed amended to the extent necessary in order that such provision be valid and enforceable, such amendment to apply only with respect to the operation of such Restrictive Covenant in the particular jurisdiction in which such adjudication is made. Section 19. Prior Agreements. This Agreement supersedes all prior agreements and understandings (including verbal agreements) between Executive and the Company regarding the terms and conditions of Executive’s employment with the Company. Section 20. 409A Compliance. (a) It is intended that each installment of the payments and benefits provided under Section 7 of this Agreement shall be treated as a separate “payment” for purposes of Section 409A of the United States Internal Revenue Code of 1986, as amended (the “Code”). (b) Notwithstanding anything contained herein to the contrary, the Executive shall not be considered to have terminated employment with the Company for purposes of Section 7 and 8 of this Agreement unless she would be considered to have incurred a “separation from service” from the Company within the meaning of Treasury Regulation §1.409A-1(h)(1)(ii). (c) The severance payments set forth in Section 8(a)(i) of this Agreement are intended to comply with the involuntary separation pay plan exception to Section 409A set forth in Treas. Reg. §1.409A-1(b)(9)(iii) to the extent that the payments due under Section 8(a) are in the aggregate less than the lesser of two times the lesser of (i) the sum of the Executive’s annualized compensation based upon the annual rate of pay for services provided to the Company for the taxable year of the Company preceding the taxable year in which the Termination Date occurs (adjusted for any increase during that year that was expected to continue indefinitely if Executive had not separated from service) and (ii) the applicable compensation limit under Section 401(a)(17) of the Code for the year in which the Termination Date occurs shall be in accordance with as an involuntary separation plan.

13 (d) The payment set forth in Section 8(a)(iv) of this Agreement is intended to comply with the short-term deferral exception to Section 409A of the Code set forth in Treas. Reg. §1.409A-1(b)(4). (e) All references in this Agreement to Executive’s termination of employment shall mean Executive’s “separation from service” within the meaning of Section 409A of the Code and Treas. Reg. §1.409A-1(h). (f) Executive acknowledges and agrees that notwithstanding any provision of this Agreement, the Company and its affiliates are not providing Executive with any tax advice with respect to Section 409A of the Code or otherwise and are not making any guarantees or other assurances of any kind to Executive with respect to the tax consequences or treatment of any amounts paid or payable to Executive under this Agreement. (g) Notwithstanding anything to the contrary contained herein, if necessary to comply with the restriction in Section 409A(a)(2)(B) of the Code concerning payments to “specified Executives,” any payment on account of the Executive’s separation from service that would otherwise be due hereunder within six (6) months after such separation shall nonetheless be delayed until the first business day of the seventh month following the Executive’s date of termination and the first such payment shall include the cumulative amount of any payments that would have been paid prior to such date if not for such restriction, together with interest on such cumulative amount during the period of such restriction at a rate, per annum, equal to the applicable federal short-term rate (compounded monthly) in effect under Section 1274(d) of the Code on the date of termination. If the Executive dies during the six-month postponement period prior to the payment, the amount of the payment deferred on account of Section 409A of the Code shall be paid to the personal representative of the Executive’s estate within thirty (30) days after the date of the Executive’s death. For purposes of Section 8 hereof, the Executive shall be a “specified Executive” for the 12-month period beginning on the first day of the fourth month following each “Identification Date” if he is a “key Executive” (as defined in Section 416(i) of the Code without regard to Section 416(i)(5) thereof) of the Company at any time during the 12-month period ending on the “Identification Date.” For purposes of the foregoing, the Identification Date shall be December 31. (h) To the extent that any provision in this Agreement is ambiguous as to its compliance with Section 409A, the provision shall be read in such a manner so that no payments due under this Agreement shall be subject to an “additional tax” as defined in Section 409A(a)(1)(B) of the Code. (i) All reimbursements provided under this Agreement shall be made or provided in accordance with the requirements of Section 409A, including, where applicable, the requirement that (i) any reimbursement is for expenses incurred during the Executive’s lifetime (or during a shorter period of time specified in this Agreement), (ii) the amount of expenses eligible for reimbursement during a calendar year may not affect the expenses eligible for reimbursement in any other calendar year, (iii) the reimbursement of an eligible expense will be made on or before the last day of the calendar year following the year in which the expense is incurred, and (iv) the right to reimbursement is not subject to liquidation or exchange for another benefit.

14 (j) In no event whatsoever shall the Company be liable for any additional tax, interest or penalty that may be imposed on Executive by Section 409A or damages for failing to comply with Section 409A. Section 21. Section 280G Limitation. If any payment(s) or benefit(s) the Executive would receive pursuant to this Agreement and/or pursuant to any other agreement or arrangement would (a) constitute a “parachute payment” within the meaning of Section 280G of the Code, (b) but for this Section, be subject to the excise tax imposed by Section 4999 of the Code (the “Excise Tax”), and if the net-after tax amount (taking into account all applicable taxes payable by the Executive, including any Excise Tax) that the Executive would receive with respect to such payments or benefits does not exceed the Reduced Amount, then such payment(s) or benefit(s) (collectively, “Payments”) shall be reduced to the Reduced Amount. The “Reduced Amount” shall be the largest portion of the Payments that can be paid or provided without causing any portion of the Payments being subject to the Excise Tax. If a reduction in payments or benefits constituting “parachute payments” is necessary so that the Payments equal the Reduced Amount, reduction shall occur in the following order: (i) first, any base salary severance payments (with payments paid latest in time reduced in first); (ii) second, any other cash payments due under any other agreement between the Company and the Executive; (iii) third, cancellation of the acceleration of vesting of any stock options; (iv) fourth, cancellation of the acceleration of vesting of any restricted stock and restricted stock units; and (v) lastly, other non-cash forms of benefits. Calculations of the foregoing will be performed at the expense of the Company by an accounting firm selected by the Company. The determinations of such accounting firm shall be final, binding and conclusive upon the Company and the Executive. IN WITNESS WHEREOF, the Company has caused this Agreement to be executed by their respective duly authorized officers and the Executive has signed this Agreement. all as of the first date above written but effective as of the Effective Date. /s/ Kristen K. Buck, M.D. Kristen K. Buck, M.D. /s/ David J. Mazzo David J. Mazzo, Ph.D. President & CEO, Lisata Therapeutics, Inc.

15 Exhibit A to Employment Agreement Employee Confidentiality, Non-Compete and Inventions Assignment Agreement I (the “Employee”) recognize that Lisata Therapeutics, Inc., a Delaware corporation (the “Company”), is a clinical-stage biopharmaceutical company dedicated to the development and commercialization of cellular therapies designed to reverse disease and/or promote the regeneration of damaged tissue. The Company is developing first-in-class therapeutics based on the characteristics of naturally occurring CD34+ cells and their ability to stimulate the growth of new microvasculature. Its technology leverages these cells to enable the body’s natural repair mechanisms using formulations unique to each medical indication (the “Business”). The “Business” also includes any other regenerative medicine, cellular therapies or other therapeutic developments initiatives that are or become a part of the Company’s (or its subsidiaries’) business during my employment tenure with the Company. Any company with which the Company enters into, or seeks or considers entering into, a business relationship in furtherance of the Business is referred to as a “Business Partner.” I understand that as part of my performance of duties as an employee of the Company (the “Engagement”), I will have access to confidential or proprietary information of the Company and the Business Partners, and I may make new contributions and inventions of value to the Company. I further understand that my Engagement creates in me a duty of trust and confidentiality to the Company with respect to any information: (1) related, applicable or useful to the business of the Company, including the Company’s anticipated research and development or such activities of its Business Partners; (2) resulting from tasks performed by me for the Company; (3) resulting from the use of equipment, supplies or facilities owned, leased or contracted for by the Company; or (4) related, applicable or useful to the business of any partner, client or customer of the Company, which may be made known to me or learned by me during the period of my Engagement. For purposes of this Agreement, the following definitions apply: “Proprietary Information” shall mean information relating to the Business or the business of any Business Partner and generally unavailable to the public that has been created, discovered, developed or otherwise has become known to the Company or in which property rights have been assigned or otherwise conveyed to the Company or a Business Partner, which information has economic value or potential economic value to the business in which the Company is or will be engaged. Proprietary Information shall include, but not be limited to, trade secrets, processes, formulas, writings, data, know-how, negative know-how, improvements, discoveries, developments, designs, inventions, techniques, technical data, patent applications, customer and supplier lists, financial information, business plans or projections and any modifications or enhancements to any of the above. “Inventions” shall mean all Business-related discoveries, developments, designs, improvements, inventions, formulas, software programs, processes, techniques, know-how, writings, graphics and other data, whether or not patentable or registrable under patent, copyright or similar statutes, that are related to or useful in the business or future business of the Company or its Business Partners or result from use of premises or other property owned, leased or contracted for by the Company. Without limiting the generality of the foregoing, Inventions shall

16 also include anything related to the Business that derives actual or potential economic value from not being generally known to the public or to other persons who can obtain economic value from its disclosure or use. As part of the consideration for my Engagement pursuant to the terms of the employment agreement between the Company and me effective as of the Start Date (the “Employment Agreement”), and the base salary, stock options, RSUs and/or other compensation and benefits to be received by me from the Company pursuant to the Employment Agreement, I hereby agree as follows: 1. Proprietary Information and Inventions. The Company, its Business Partners or their respective assigns, as the case may be, are and shall be the sole owner of all Proprietary Information and Inventions related to the Business and the sole owner of all patents, trademarks, service marks, copyrights, mask rights and other rights (collectively referred to herein as “Rights”) pertaining to any Proprietary Information or Inventions. I hereby acknowledge that all original works of authorship that are made by me (solely or jointly with others) within the scope of my Engagement and which are protectable by copyright are “works for hire” as that term is defined in the United States Copyright Act (17 USCA, Section 101). I further hereby assign to the Company, any Rights I may have or acquire in any Proprietary Information or Inventions which arise in the course of my Engagement. I further agree to assist the Company or any person designated by it in every proper way (but at the Company’s expense) to obtain and from time to time enforce Rights relating to said Proprietary Information or Inventions in any and all countries. I will execute all documents for use in applying for, obtaining and enforcing such Rights in such Proprietary Information or Inventions as the Company may desire, together with any assignments thereof to the Company or persons designated by it. My obligation to assist the Company or any person designated by it in obtaining and enforcing Rights relating to Proprietary Information or Inventions shall continue beyond the cessation of my Engagement (“Cessation of my Engagement”). In the event the Company is unable, after reasonable effort, to secure my signature on any document or documents needed to apply for or enforce any Right relating to Proprietary Information or to an Invention, whether because of my physical or mental incapacity or for any other reason whatsoever, I hereby irrevocably designate and appoint the Company and its duly authorized officers and agents as my agents and attorneys-in-fact to act for and in my behalf and stead in the execution and filing of any such application and in furthering the application for and enforcement of Rights with the same legal force and effect as if such acts were performed by me. 2. Confidentiality. At all times, both during my Engagement and after the Cessation of my Engagement, whether the cessation is voluntary or involuntary, for any reason or no reason, or by disability, I will keep in strictest confidence and trust all Proprietary Information, and I will not disclose or use or permit the use or disclosure of any Proprietary Information or Rights pertaining to Proprietary Information, or anything related thereto, without the prior written consent of the Company, except as may be necessary in the ordinary course of performing my duties for the Company or to enforce any of my rights under my Employment Agreement. I recognize that the Company has received and in the future will receive from third parties (including Business Partners) their confidential or proprietary information subject to a duty on the Company’s part to maintain the confidentiality of such information and to use it only for certain limited purposes. I agree that I owe the Company and such third parties (including Business Partners), during my Engagement and after the Cessation of my Engagement, a duty to hold all such confidential or

17 proprietary information in the strictest confidence, and I will not disclose or use or permit the use or disclosure of any such confidential or proprietary information without the prior written consent of the Company, except as may be necessary in the ordinary course of performing my duties for the Company consistent with the Company’s agreement with such third party or to enforce any of my rights under my Employment Agreement or otherwise. 3. Noncompetition and Nonsolicitation. (a) During my Engagement, and for a period of two (2) years after the Cessation of my Engagement, I will not directly or indirectly, whether alone or in concert with others or as a partner, officer, director, consultant, agent, employee or stockholder of any company or commercial enterprise, engage in any activity in the United States, Canada, Asia and Europe that is in Competition with the Company concerning its work or any Business Partner’s work in the Business. Further during my Engagement and for a period of two (2) years after the Cessation of my Engagement, I agree not to plan or otherwise take any preliminary steps, either alone or in concert with others to set up or reengage in any business enterprise that would be in Competition with the Company in the Business: provided, however, that the foregoing shall not restrict my ability to seek other employment following the Cessation of my Engagement (whether or riot I am receiving or have received any Additional Payments) as long as I do not actually commence such employment. In addition, nothing in this Agreement shall preclude me from providing services to an entity which operates multiple businesses including indirectly, through its affiliates, a business that is in Competition with the Company’s Business, if that competitive affiliate is not a material part of the business of such entity and if I do not provide services, directly or indirectly, with respect to, or have supervisory or executive authority with respect to, any such affiliate which is itself directly engaged in such business that is in Competition with the Company’s work in the Business. For purposes of this Agreement, “Competition” shall mean any involvement in any project competitive with the Business (as defined above) or the therapies being developed as part of the Business. (b) During my Engagement and for a period of two (2) years after the Cessation of my Engagement, I will not directly or indirectly, whether alone or in concert with others or as a partner, officer, director, consultant, agent, employee or stockholder of any company or commercial enterprise, either alone or in concert with others, not take any of the following actions: (i) persuade or attempt to persuade any Business Partner, Customer, Prospective Customer or Supplier to cease doing business with the Company, or to reduce the amount of business it does with the Company; (ii) persuade or attempt to persuade any Service Provider to cease providing services to the Company or any Business Partner; or (iii) solicit for hire or hire for myself or for any Person any Service Provider The following definitions are applicable to this Section 3(b): (A) “Customer” means any Person that purchased goods or services from the Company, or engages in a collaborative arrangement with the Company, at any time within I year prior to the date of the solicitation prohibited by Section 3(b)(i) or (ii).

18 (B) “Prospective Customer” means any Person with whom the Company met or to whom the Company presented for the purpose of soliciting the Person to become a Customer of the Company within 6 months prior to the date of the solicitation prohibited by Section 3(b)(i) or (ii). (C) “Service Provider” means any Person who is an employee or independent contractor of the Company or the Company or who was within twelve (12) months preceding the solicitation prohibited by Section 3(b)(iii) or (iv) an employee or independent contractor of the Company or the Company. (D) “Supplier” means any Person that sold goods or services to the Company, or engages in a collaborative arrangement with the Company at any time within twelve (12) months prior to the date of the solicitation prohibited by Section 3(b)(i) or (ii). (E) “Person” means an individual, a sole proprietorship, a corporation, a limited liability company, a partnership, an association, a trust, or other business entity, whether or not incorporated. (c) The following shall not be deemed to breach the foregoing obligations: my ownership of stock, partnership interests or other securities of any entity not in excess of two percent (2%) of any class of such interests or securities that are publicly traded. (d) Employee acknowledges that (i) the restrictions contained in this section are reasonable and necessary to protect the legitimate business interests of the Company, (ii) that the term of this obligation is reasonable in scope, and (iii) that this obligation is a material term, without which the Company would be unwilling to enter into an employment relationship with the Employee. 4. Delivery of Company Property and Work Product. In the event of the Cessation of my Engagement, I will deliver to the Company all biological materials, devices, records, sketches, reports, memoranda, notes, proposals, lists, correspondence, equipment, documents, photographs, photostats, negatives, undeveloped film, drawings, specifications, tape recordings or other electronic recordings, programs, data, marketing material and other materials or property of any nature belonging to the Company or its clients or customers, and I will not take with me, or allow a third party to take, any of the foregoing or any reproduction of any of the foregoing. 5. No Conflict. I represent, warrant and covenant that my performance of all the terms of this Agreement and the performance of my duties for the Company does not and will not breach any agreement to keep in confidence proprietary information acquired by me in confidence or in trust prior to my Engagement. I have not entered into, and I agree that I will not enter into, any agreement, either written or oral, in conflict herewith. 6. No Use of Confidential Information. I represent, warrant and covenant that I have not brought and will not bring with me to the Company or use in my Engagement any materials or documents of a former employer, or any person or entity for which I have acted as an independent contractor or consultant, that are not generally available to the public, unless I have obtained written authorization from any such former employer, person or firm for their possession and use.

19 I understand and agree that, in my service to the Company, I am not to breach any obligation of confidentiality that I have to former employers or other persons. 7. Enforcement; Equitable Relief. Employee acknowledges that any breach or threatened breach by Employee of any provision of this Agreement may result in immediate and irreparable injury to the Company, and that such injury may not be readily compensable by monetary damages. In the event of any such breach or threatened breach, Employee acknowledges that, in addition to all other remedies available at law and equity, the Company shall be entitled to seek equitable relief (including a temporary restraining order, a preliminary injunction and/or a permanent injunction), and an equitable accounting of all earnings, profits or other benefits arising from such breach and will be entitled to receive such other damages, director consequential, as may be appropriate. In addition, and not instead of, those rights, Employee further acknowledges that Employee shall be responsible for payment of the fees and expenses of the Company’s attorneys and experts, as well as the Company’s court costs, pertaining to any suit, action, or other proceeding, arising directly or indirectly out of Employee’s violation or threatened violation of any of the provisions of this section. The Company shall not be required to post any bond or other security in connection with any proceeding to enforce this section. 8. Severability. If any provision of this Agreement shall be determined by any court of competent jurisdiction to be unenforceable or otherwise invalid as written, the same shall be enforced and validated to the extent permitted by law. All provisions of this Agreement are severable, and the unenforceability or invalidity of any single provision hereof shall not affect the remaining provisions. 9. Miscellaneous. This Agreement shall be governed by and construed under the laws of the State of New Jersey applied to contracts made and performed wholly within such state. No implied waiver of any provision within this Agreement shall arise in the absence of a waiver in writing, and no waiver with respect to a specific circumstance, event or occasion shall be construed as a continuing waiver as to similar circumstances, events or occasions. This Agreement, together with the employment agreement between the Company and myself, contains the sole and entire agreement and understanding between the Company and myself with respect to the subject matter hereof and supersedes and replaces any prior agreements to the extent any such agreement is inconsistent herewith. This Agreement can be amended, modified, released or changed in whole or in part only by a written agreement executed by the Company and myself. This Agreement shall be binding upon me, my heirs, executors, assigns and administrators, and it shall inure to the benefit of the Company and each of its successors or assigns. This Agreement shall be effective as of the first day of my being retained to render services to the Company, even if such date precedes the date I sign this Agreement. 10. Thorough Understanding of Agreement. I have read all of this Agreement and understand it completely, and by my signature below I represent that this Agreement is the only statement made by or on behalf of the Company upon which I have relied in signing this Agreement.

20 IN WITNESS WHEREOF, I have caused this Employee Confidentiality, Non-Compete and Inventions Assignment Agreement to be signed on the date written below. DATED: June 10, 2025 Kristen K. Buck, MD /s/ Kristen K. Buck, M.D.

Exhibit 10.3 AMENDED AND RESTATED SEPARATION BENEFITS AGREEMENT This SEPARATION BENEFITS AGREEMENT (this “Agreement”), dated as of June 10, 2025, is by and between Lisata Therapeutics Inc. (the “Company”) and James Nisco (“Executive”). WITNESSETH WHEREAS, the Company and Executive previously entered into the Prior Agreements (as defined in Section 1(f) below); and WHEREAS the Company desires to enter in an agreement setting forth the mutual understandings and agreements reached between the Company and Executive with respect to Executive’s benefits upon a separation of employment with the Company, as set forth herein. NOW, THEREFORE, in consideration of the promises and the mutual covenants and agreements contained herein and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto, intending to be legally bound hereby, agree as follows: Section 1. Definitions. As used herein the following terms shall have the following meanings: (a) “Cause” shall mean that Executive has: (i) committed gross negligence in connection with Executive’s duties or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (ii) committed fraud in connection with Executive’s duties or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (iii) engaged in personal dishonesty, willful misconduct, willful violation of any law, or breach of fiduciary duty, in each instance, with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (iv) been indicted for, or has been found by a court of competent jurisdiction to have committed or pled guilty to, (A) a felony (or state law equivalent) or (B) any other serious crime involving moral turpitude or that has (or is reasonably likely to have) a material adverse effect either on (x) Executive’s ability to perform Executive’s duties for the Company or (y) the reputation and goodwill of the Company, regardless of whether or not such other crime is related or unrelated to the business of the Company, its subsidiaries or other affiliates; (v) shown chronic use of alcohol, drugs or other similar substances that materially affects Executive’s work performance; (vi) breached Executive’s obligations under any written agreement between Executive and the Company related to confidentiality, non-competition, non-solicitation or the assignment of intellectual property; (vii) failed to materially perform Executive’s duties or to follow the lawful directives of the Company; provided, that, if such failure described in this clause (vii) is susceptible to cure (as determined in the reasonable discretion of the Company’s Board of Directors (the “Board”)), Executive shall have thirty (30) days after notice from the Company to cure such failure; or (viii) materially violated the Company’s written code of conduct or other written or established policies and/or procedures in place from time to time; provided, that, if such violation described in this clause (viii) is susceptible to cure (as determined in the reasonable discretion of the Board), Executive

shall have thirty (30) days after notice from the Company to cure such violation. Any notice to Executive under this Section 1(b) shall be in writing and shall specify in reasonable detail Executive’s acts or omissions that the Company alleges constitute “Cause.” (b) “Change in Control” shall mean a transaction or a series of related transactions in which: (i) all or substantially all of the assets of the Company are transferred to any “person” or “group” (as such terms are defined in Section 13(d)(3) and 14(d)(2) of the Exchange Act), other than the sale of all or substantially all of the assets of the Company to a person or group who beneficially own, directly or indirectly, at least fifty percent (50%) or more of the combined voting power of the outstanding voting securities of the Company at the time of such sale; (ii) any person or group becomes the “beneficial owner” (as defined in Rules 13d-3 and 13d-5 under the Exchange Act), directly or indirectly, of the Company’s outstanding equity representing more than thirty percent (30%) of the total voting power of the Company’s then-outstanding equity; (iii) the Company undergoes a merger, reorganization or other consolidation in which the holders of the outstanding equity of the Company immediately prior to such merger, reorganization or consolidation own less than fifty percent (50%) of the surviving entity’s voting power immediately after the transaction; (iv) the date a majority of the members of the Company’s incumbent Board is replaced during any twelve (12) month period by members whose appointment or election is not endorsed by a majority to the Company’s incumbent Board before the date of the appointment or election, or (v) the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided further that the Change in Control meets all of the requirements of a “change in the ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(v), a “change in the effective ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(vi), or “a change in the ownership of a substantial portion of the corporation’s assets” within in the meaning of Treasury Regulation §1.409A-3(i)(5)(vii). (c) “Disability” shall mean that Executive has been unable to perform Executive’s duties to the Company on account of physical or mental illness or incapacity for a period of ninety (90) consecutive calendar days, or one hundred twenty (120) calendar days (whether or not consecutive) during any three hundred sixty five- (365-) day period, as a result of a condition that is treated as a total or permanent disability under the long-term insurance policy of the Company that covers Executive. (d) “Good Reason” shall mean (i) a material reduction in Executive’s base salary; (ii) Executive’s position, duties, responsibilities, or authority have been materially reduced or Executive has been repeatedly assigned duties that are materially inconsistent with Executive’s duties, in each case, without Executive’s consent; or (iii) the requirement that Executive relocate Executive’s primary place of employment more than fifty (50) miles from Executive’s current place of employment (unless such location is closer to Executive’s primary residence). “Good Reason” shall not be deemed to exist, however, unless (x) Executive has given written notice to the Company specifying in reasonable detail the Company’s acts or omissions that Executive alleges constitutes “Good Reason” within sixty (60) days after the first occurrence of such circumstances and the Company shall have failed to cure any such act or omission within sixty (60) days of receipt of such written notice, and (y) Executive actually terminates Executive’s employment within one hundred eighty (180) days following the initial occurrence of the condition Executive considers to be “Good Reason.” If Executive fails to provide this notice and cure period

prior to Executive’s resignation or resigns more than one hundred eighty (180) days after the initial existence of the condition, Executive’s resignation will not be deemed to be for “Good Reason.” (e) “Prior Agreements” shall mean the Change in Control Letter Agreement between Executive and Caladrius Biosciences (the Company’s predecessor) dated as of September 12, 2016 and Revised as of March 25, 2022 and the Non-Change in Control (CIC)* Separation Pay Agreement between the Company and Executive, dated as of December 24, 2025. Section 2. Separation Benefits. If the Company terminates Executive’s employment without Cause (other than by reason of Executive’s death or Disability) or Executive terminates Executive’s employment for Good Reason at any time prior to the effective date of a Change in Control, subject to Executive’s execution of the Release (as defined in Section 4 below), the Company will: (a) Pay the Executive a lump-sum amount, within thirty (30) days following the date the termination of Executive’s employment becomes effective (the “Termination Date”) (or earlier if required by law), equal to the sum of (i) his earned but unpaid Base Salary through (ii) any bonus amount earned and vested but not paid for periods ending on or prior to the Termination Date, (iii) any accrued and unused Paid Time Off (“PTO”) per the PTO Policy, (iv) any unreimbursed business expenses or other amounts due to the Executive from the Company as of the Termination Date, and (v) all other payments and benefits to which the Executive then may be entitled under the terms of any applicable compensation arrangement or benefit, equity or perquisite plan or program or grant, including but not limited to any applicable insurance benefits; (b) Continue to pay Executive’s current base salary for a period beginning on the Termination Date and ending on the twelve (12) month anniversary of the Termination Date (the “Severance Period”), commencing on the next payroll period following the effective date of the Release (subject to Executive’s rights set forth in Section 4 below); (c) Pay Executive a lump amount equal to 100% of Executive’s then annual target bonus on the next payroll period following the effective date of the Release; (d) If Executive then participates in the Company’s medical and/or dental plans and Executive timely elects to continue and maintain group health plan coverage pursuant to the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), reimburse Executive monthly for the duration of the Severance Period, the cost of such coverage for the Severance Period, which payments will be equal to the amount of the monthly premium for such coverage (the “COBRA Assistance”); provided, however, if the Company is unable to provide the COBRA Assistance without incurring tax penalties or violating applicable nondiscrimination requirements or the Company terminates the health plans in which Executive is participating and no other health plan is required to provide the COBRA Assistance to Executive, then in lieu of the forgoing benefit, the Company will pay Executive a lump sum cash payment equal to the aggregate cost of the remaining Severance Period for which benefits were to be reimbursed, less any required withholdings within ten (10) business days after Executive’s participation in the health plan terminates; and

(e) extend the exercise date of any vested options to the earlier of the one-year anniversary of the Termination Date and the original expiration date of the options (the foregoing (b) through (e), the “Separation Benefits”). Section 3. Additional Change in Control Separation Benefits. If the Company terminates Executive's employment without Cause (other than by reason of death or Disability) or the Executive voluntarily terminates his employment for Good Reason during the period commencing on the effective date of a Change in Control and ending on the second anniversary of the effective date of a Change in Control, in addition to the Separation Benefits set forth in Section 2, the Company will fully vest as of the Termination Date all outstanding unvested equity awards, including options granted to the Executive. Section 4. Payment; Release. (a) Notwithstanding anything in Section 2 to the contrary, and subject to Section 5 below, prior to the date the first payment is made under Section 2, Executive may elect to receive a portion of the aggregate amount to which Executive is eligible to receive under Section 2(b) equal to (a) the product of the compensation limit under Section 401(a)(17), and (b) two (2), in a lump sum payment on the next payroll period following the Termination Date. For the avoidance of doubt, any payment made pursuant to this Section 3 shall be considered a right to receive a series of separate payments (the “First Separation Benefit Plan”), and Executive shall continue to be eligible to receive any amounts pursuant to Section 2(b) in accordance with the terms of Section 2(b), less any amounts paid under the First Separation Benefit Plan. (b) Executive’s right to the payments and benefits described in Section 2 and Section 3 is conditioned upon Executive’s executing and not revoking a valid separation and general release agreement in a form provided by the Company (the “Release”), and provided such release becomes effective and irrevocable within sixty (60) days following termination or such shorter time period set forth therein, releasing the Company, its subsidiaries, other affiliates and shareholders from any and all liability. Any payments or benefits due for the period after termination and before the Release becomes effective shall be paid with the first payment after the Release becomes effective. Notwithstanding any other provision herein, if the period during which Executive has discretion to execute or revoke the Release straddles two (2) calendar years, the Company shall make payments conditioned on the Release no earlier than January 1st of the second (2nd) calendar year, regardless of the year in which the Release becomes effective. Section 5. Interpretation. The Board will determine in its reasonable discretion whether a termination of Executive’s employment would entitle Executive to the severance benefits set forth in Sections 2 and 3. If the Company consummates a Change in Control, the members of the Board as constituted immediately prior to the closing of a Change in Control (the “Incumbent Board”) shall retain authority to make the forgoing determinations concerning the termination of Executive’s employment until the second (2nd) anniversary of the closing date of the Change in Control regardless of whether the Company’s Board is reconstituted in connection with the Change in Control. Following a Change in Control, the Incumbent Board may delegate its authority under this Section 5 to a qualified independent third party, such as a retired judge selected by the head of the American Arbitration Association in Manhattan, an independent compensation consultant or a law firm

Section 6. Section 409A. (a) It is intended that each installment of the payments and benefits provided under Sections 2 and 3 of this Agreement shall be treated as a separate “payment” for purposes of Section 409A of the United States Internal Revenue Code of 1986, as amended (the “Code”). (b) Notwithstanding anything contained herein to the contrary, the Executive shall not be considered to have terminated employment with the Company for purposes of Section 6 and 7 of this Agreement unless he would be considered to have incurred a “separation from service” from the Company within the meaning of Treasury Regulation §1.409A-1(h)(1)(ii). (c) The severance payments set forth in Section 2(b) of this Agreement are intended to comply with the involuntary separation pay plan exception to Section 409A set forth in Treas. Reg. §1.409A-1(b)(9)(iii) to the extent that the payments due under Section 2(b) are in the aggregate less than the lesser of two times the lesser of (i) the sum of the Executive’s annualized compensation based upon the annual rate of pay for services provided to the Company for the taxable year of the Company preceding the taxable year in which the Termination Date occurs (adjusted for any increase during that year that was expected to continue indefinitely if Executive had not separated from service) and (ii) the applicable compensation limit under Section 401(a)(17) of the Code for the year in which the Termination Date occurs shall be in accordance with as an involuntary separation plan. (d) The payment set forth in Section 2(c) of this Agreement is intended to comply with the short-term deferral exception to Section 409A of the Code set forth in Treas. Reg. §1.409A-1(b)(4). (e) All references in this Agreement to Executive’s termination of employment shall mean Executive’s “separation from service” within the meaning of Section 409A of the Code and Treas. Reg. §1.409A-1(h). (f) Executive acknowledges and agrees that notwithstanding any provision of this Agreement, the Company and its affiliates are not providing Executive with any tax advice with respect to Section 409A of the Code or otherwise and are not making any guarantees or other assurances of any kind to Executive with respect to the tax consequences or treatment of any amounts paid or payable to Executive under this Agreement. (g) Notwithstanding anything to the contrary contained herein, if necessary to comply with the restriction in Section 409A(a)(2)(B) of the Internal Revenue Code of 1986, as amended (the “Code”) concerning payments to “specified Executives,” any payment on account of the Executive’s separation from service that would otherwise be due hereunder within six (6) months after such separation shall nonetheless be delayed until the first business day of the seventh month following the Executive’s date of termination and the first such payment shall include the cumulative amount of any payments that would have been paid prior to such date if not for such restriction, together with interest on such cumulative amount during the period of such restriction at a rate, per annum, equal to the applicable federal short-term rate (compounded monthly) in effect under Section 1274(d) of the Code on the date of termination. If the Executive dies during the six- month postponement period prior to the payment, the amount of the payment deferred on account

of Section 409A of the Code shall be paid to the personal representative of the Executive’s estate within 30 days after the date of the Executive’s death. For purposes of this Section 5, the Executive shall be a “specified Executive” for the 12-month period beginning on the first day of the fourth month following each “Identification Date” if he is a “key Executive” (as defined in Section 416(i) of the Code without regard to Section 416(i)(5) thereof) of the Company at any time during the 12-month period ending on the “Identification Date.” For purposes of the foregoing, the Identification Date shall be December 31. (h) To the extent that any provision in this Agreement is ambiguous as to its compliance with Section 409A, the provision shall be read in such a manner so that no payments due under this Agreement shall be subject to an “additional tax” as defined in Section 409A(a)(1)(B) of the Code. (i) In no event whatsoever shall the Company be liable for any additional tax, interest or penalty that may be imposed on Executive by Section 409A or damages for failing to comply with Section 409A. Section 6. Section 280G Limitation. If any payment(s) or benefit(s) Executive would receive pursuant to this Agreement and/or pursuant to any other agreement or arrangement would (a) constitute a “parachute payment” within the meaning of Section 280G of the Code, (b) but for this Section 5, be subject to the excise tax imposed by Section 4999 of the Code (the “Excise Tax”), and (c) if the net-after tax amount (taking into account all applicable taxes payable by Executive, including any Excise Tax) that Executive would receive with respect to such payments or benefits does not exceed the Reduced Amount, then such payment(s) or benefit(s) (collectively, “Payments”) shall be reduced to the Reduced Amount. The “Reduced Amount” shall be the largest portion of the Payments that can be paid or provided without causing any portion of the Payments being subject to the Excise Tax. If a reduction in payments or benefits constituting “parachute payments” is necessary so that the Payments equal the Reduced Amount, reduction shall occur in the following order: (i) first, any base salary severance payments; (ii) second, any other cash payments due under any other agreement between the Company and Executive; (iii) third, cancellation of the acceleration of vesting of any stock options; (iv) fourth, cancellation of the acceleration of vesting of any restricted stock and restricted stock units; and (v) lastly, other non-cash forms of benefits. Calculations of the foregoing will be performed at the expense of the Company by an accounting firm selected by the Company. The determinations of such accounting firm shall be final, binding and conclusive upon the Company and Executive. Section 7. Severance Plan. For the avoidance of doubt, Executive’s severance rights arise solely under this Agreement. Executive is not a “Participant” in Lisata Therapeutics, Inc. Severance Plan, effective June 10, 2025 (the “Severance Plan”) and is not entitled to any benefits under the Severance Plan. Section 8. Binding Agreement; No Assignment. This Agreement shall be binding upon, and shall inure to the benefit of, Executive and the Company and their respective permitted successors, assigns, heirs, beneficiaries and representatives. This Agreement is personal to Executive and may not be assigned by Executive. This Agreement may not be assigned by the Company except in connection with a sale of all or substantially all of its assets or a merger or

consolidation of the Company, and the acquiring Company or entity expressly assumes this Agreement. Any attempted assignment in violation of this Section 8 shall be null and void. Section 9. Governing Law; Consent to Jurisdiction. The validity, interpretation, performance, and enforcement of this Agreement shall be governed by the laws of the State of New Jersey. In addition, Executive and the Company irrevocably submit to the exclusive jurisdiction of the courts of the State of New Jersey and the United States District Court sitting in New Jersey for the purpose of any suit, action, proceeding or judgment relating to or arising out of this Agreement and the transactions contemplated hereby. Service of process in connection with any such suit, action or proceeding may be served on Executive or the Company anywhere in the world by the same methods as are specified for the giving of notices under this Agreement. Executive and the Company irrevocably consent to the jurisdiction of any such court in any such suit, action or proceeding and to the laying of venue in such court. In any such action or proceeding, the court shall have the authority to award reasonable costs, expenses, and attorneys’ fees to the party that substantially prevails. Section 10. Entire Agreement; Amendments. This Agreement embodies the entire agreement between Executive and the Company with respect to the subject matter hereof and may only be amended or otherwise modified by a writing executed by all of the parties hereto. This Agreement supersedes all prior agreements and understandings, written or verbal, between Executive and the Company regarding the terms and conditions of the subject matter hereof, including the Prior Agreements. Section 11. Counterparts. This Agreement may be executed in any number of counterparts, each of which when executed shall be deemed to be an original and all of which together shall be deemed to be one and the same instrument. Section 12. Severability. If any provision, section or paragraph, or specific term contained therein, of this Agreement or the application thereof is determined by a court to be illegal, invalid or unenforceable, that provision, section, paragraph or term shall not be a part of this Agreement, and the legality, validity and enforceability of remaining provisions, sections and paragraphs, and all other terms therein, of this Agreement shall not be affected thereby. [Signature Page Follows]

LISATA THERAPEUTICS, INC. By: /s/ David J. Mazzo Name: David J. Mazzo, Ph.D. President and Chief Executive Officer Agreed to and Accepted: /s/ James Nisco Date: June 10, 2025

Exhibit 10.4 AMENDED AND RESTATED SEPARATION BENEFITS AGREEMENT This SEPARATION BENEFITS AGREEMENT (this “Agreement”), dated as of June 10, 2025, is by and between Lisata Therapeutics Inc. (the “Company”) and Tariq Imam (“Executive”). WITNESSETH WHEREAS, the Company and Executive previously entered into the Prior Agreements (as defined in Section 1(f) below); and WHEREAS the Company desires to enter in an agreement setting forth the mutual understandings and agreements reached between the Company and Executive with respect to Executive’s benefits upon a separation of employment with the Company, as set forth herein. NOW, THEREFORE, in consideration of the promises and the mutual covenants and agreements contained herein and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto, intending to be legally bound hereby, agree as follows: Section 1. Definitions. As used herein the following terms shall have the following meanings: (a) “Cause” shall mean that Executive has: (i) committed gross negligence in connection with Executive’s duties or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (ii) committed fraud in connection with Executive’s duties or otherwise with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (iii) engaged in personal dishonesty, willful misconduct, willful violation of any law, or breach of fiduciary duty, in each instance, with respect to the business and affairs of the Company, its subsidiaries and/or its other affiliates; (iv) been indicted for, or has been found by a court of competent jurisdiction to have committed or pled guilty to, (A) a felony (or state law equivalent) or (B) any other serious crime involving moral turpitude or that has (or is reasonably likely to have) a material adverse effect either on (x) Executive’s ability to perform Executive’s duties for the Company or (y) the reputation and goodwill of the Company, regardless of whether or not such other crime is related or unrelated to the business of the Company, its subsidiaries or other affiliates; (v) shown chronic use of alcohol, drugs or other similar substances that materially affects Executive’s work performance; (vi) breached Executive’s obligations under any written agreement between Executive and the Company related to confidentiality, non-competition, non-solicitation or the assignment of intellectual property; (vii) failed to materially perform Executive’s duties or to follow the lawful directives of the Company; provided, that, if such failure described in this clause (vii) is susceptible to cure (as determined in the reasonable discretion of the Company’s Board of Directors (the “Board”)), Executive shall have thirty (30) days after notice from the Company to cure such failure; or (viii) materially violated the Company’s written code of conduct or other written or established policies and/or procedures in place from time to time; provided, that, if such violation described in this clause (viii) is susceptible to cure (as determined in the reasonable discretion of the Board), Executive

shall have thirty (30) days after notice from the Company to cure such violation. Any notice to Executive under this Section 1(b) shall be in writing and shall specify in reasonable detail Executive’s acts or omissions that the Company alleges constitute “Cause.” (b) “Change in Control” shall mean a transaction or a series of related transactions in which: (i) all or substantially all of the assets of the Company are transferred to any “person” or “group” (as such terms are defined in Section 13(d)(3) and 14(d)(2) of the Exchange Act), other than the sale of all or substantially all of the assets of the Company to a person or group who beneficially own, directly or indirectly, at least fifty percent (50%) or more of the combined voting power of the outstanding voting securities of the Company at the time of such sale; (ii) any person or group becomes the “beneficial owner” (as defined in Rules 13d-3 and 13d-5 under the Exchange Act), directly or indirectly, of the Company’s outstanding equity representing more than thirty percent (30%) of the total voting power of the Company’s then-outstanding equity; (iii) the Company undergoes a merger, reorganization or other consolidation in which the holders of the outstanding equity of the Company immediately prior to such merger, reorganization or consolidation own less than fifty percent (50%) of the surviving entity’s voting power immediately after the transaction; (iv) the date a majority of the members of the Company’s incumbent Board is replaced during any twelve (12) month period by members whose appointment or election is not endorsed by a majority to the Company’s incumbent Board before the date of the appointment or election, or (v) the approval by the shareholders of the Company of any plan of complete liquidation of the Company, provided further that the Change in Control meets all of the requirements of a “change in the ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(v), a “change in the effective ownership of a corporation” within the meaning of Treasury Regulation §1.409A-3(i)(5)(vi), or “a change in the ownership of a substantial portion of the corporation’s assets” within in the meaning of Treasury Regulation §1.409A-3(i)(5)(vii). (c) “Disability” shall mean that Executive has been unable to perform Executive’s duties to the Company on account of physical or mental illness or incapacity for a period of ninety (90) consecutive calendar days, or one hundred twenty (120) calendar days (whether or not consecutive) during any three hundred sixty five- (365-) day period, as a result of a condition that is treated as a total or permanent disability under the long-term insurance policy of the Company that covers Executive. (d) “Good Reason” shall mean (i) a material reduction in Executive’s base salary; (ii) Executive’s position, duties, responsibilities, or authority have been materially reduced or Executive has been repeatedly assigned duties that are materially inconsistent with Executive’s duties, in each case, without Executive’s consent; or (iii) the requirement that Executive relocate Executive’s primary place of employment more than fifty (50) miles from Executive’s current place of employment (unless such location is closer to Executive’s primary residence). “Good Reason” shall not be deemed to exist, however, unless (x) Executive has given written notice to the Company specifying in reasonable detail the Company’s acts or omissions that Executive alleges constitutes “Good Reason” within sixty (60) days after the first occurrence of such circumstances and the Company shall have failed to cure any such act or omission within sixty (60) days of receipt of such written notice, and (y) Executive actually terminates Executive’s employment within one hundred eighty (180) days following the initial occurrence of the condition Executive considers to be “Good Reason.” If Executive fails to provide this notice and cure period

prior to Executive’s resignation or resigns more than one hundred eighty (180) days after the initial existence of the condition, Executive’s resignation will not be deemed to be for “Good Reason.” (e) “Prior Agreements” shall mean the Change in Control Letter Agreement between Executive and Caladrius Biosciences (the Company’s predecessor) dated as of March 25, 2022 and the Non-Change in Control (CIC)* Separation Pay Agreement between the Company and Executive, dated as of December 24, 2025. Section 2. Separation Benefits. If the Company terminates Executive’s employment without Cause (other than by reason of Executive’s death or Disability) or Executive terminates Executive’s employment for Good Reason at any time prior to the effective date of a Change in Control, subject to Executive’s execution of the Release (as defined in Section 4 below), the Company will: (a) Pay the Executive a lump-sum amount, within thirty (30) days following the date the termination of Executive’s employment becomes effective (the “Termination Date”) (or earlier if required by law), equal to the sum of (i) his earned but unpaid Base Salary through (ii) any bonus amount earned and vested but not paid for periods ending on or prior to the Termination Date, (iii) any accrued and unused Paid Time Off (“PTO”) per the PTO Policy, (iv) any unreimbursed business expenses or other amounts due to the Executive from the Company as of the Termination Date, and (v) all other payments and benefits to which the Executive then may be entitled under the terms of any applicable compensation arrangement or benefit, equity or perquisite plan or program or grant, including but not limited to any applicable insurance benefits; (b) Continue to pay Executive’s current base salary for a period beginning on the Termination Date and ending on the twelve (12) month anniversary of the Termination Date (the “Severance Period”), commencing on the next payroll period following the effective date of the Release (subject to Executive’s rights set forth in Section 4 below); (c) Pay Executive a lump amount equal to 100% of Executive’s then annual target bonus on the next payroll period following the effective date of the Release; (d) If Executive then participates in the Company’s medical and/or dental plans and Executive timely elects to continue and maintain group health plan coverage pursuant to the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended (“COBRA”), reimburse Executive monthly for the duration of the Severance Period, the cost of such coverage for the Severance Period, which payments will be equal to the amount of the monthly premium for such coverage (the “COBRA Assistance”); provided, however, if the Company is unable to provide the COBRA Assistance without incurring tax penalties or violating applicable nondiscrimination requirements or the Company terminates the health plans in which Executive is participating and no other health plan is required to provide the COBRA Assistance to Executive, then in lieu of the forgoing benefit, the Company will pay Executive a lump sum cash payment equal to the aggregate cost of the remaining Severance Period for which benefits were to be reimbursed, less any required withholdings within ten (10) business days after Executive’s participation in the health plan terminates; and

(e) extend the exercise date of any vested options to the earlier of the one-year anniversary of the Termination Date and the original expiration date of the options (the foregoing (b) through (e), the “Separation Benefits”). Section 3. Additional Change in Control Separation Benefits. If the Company terminates Executive's employment without Cause (other than by reason of death or Disability) or the Executive voluntarily terminates his employment for Good Reason during the period commencing on the effective date of a Change in Control and ending on the second anniversary of the effective date of a Change in Control, in addition to the Separation Benefits set forth in Section 2, the Company will fully vest as of the Termination Date all outstanding unvested equity awards, including options granted to the Executive. Section 4. Payment; Release. (a) Notwithstanding anything in Section 2 to the contrary, and subject to Section 5 below, prior to the date the first payment is made under Section 2, Executive may elect to receive a portion of the aggregate amount to which Executive is eligible to receive under Section 2(b) equal to (a) the product of the compensation limit under Section 401(a)(17), and (b) two (2), in a lump sum payment on the next payroll period following the Termination Date. For the avoidance of doubt, any payment made pursuant to this Section 3 shall be considered a right to receive a series of separate payments (the “First Separation Benefit Plan”), and Executive shall continue to be eligible to receive any amounts pursuant to Section 2(b) in accordance with the terms of Section 2(b), less any amounts paid under the First Separation Benefit Plan. (b) Executive’s right to the payments and benefits described in Section 2 and Section 3 is conditioned upon Executive’s executing and not revoking a valid separation and general release agreement in a form provided by the Company (the “Release”), and provided such release becomes effective and irrevocable within sixty (60) days following termination or such shorter time period set forth therein, releasing the Company, its subsidiaries, other affiliates and shareholders from any and all liability. Any payments or benefits due for the period after termination and before the Release becomes effective shall be paid with the first payment after the Release becomes effective. Notwithstanding any other provision herein, if the period during which Executive has discretion to execute or revoke the Release straddles two (2) calendar years, the Company shall make payments conditioned on the Release no earlier than January 1st of the second (2nd) calendar year, regardless of the year in which the Release becomes effective. Section 5. Interpretation. The Board will determine in its reasonable discretion whether a termination of Executive’s employment would entitle Executive to the severance benefits set forth in Sections 2 and 3. If the Company consummates a Change in Control, the members of the Board as constituted immediately prior to the closing of a Change in Control (the “Incumbent Board”) shall retain authority to make the forgoing determinations concerning the termination of Executive’s employment until the second (2nd) anniversary of the closing date of the Change in Control regardless of whether the Company’s Board is reconstituted in connection with the Change in Control. Following a Change in Control, the Incumbent Board may delegate its authority under this Section 5 to a qualified independent third party, such as a retired judge selected by the head of the American Arbitration Association in Manhattan, an independent compensation consultant or a law firm

Section 6. Section 409A. (a) It is intended that each installment of the payments and benefits provided under Sections 2 and 3 of this Agreement shall be treated as a separate “payment” for purposes of Section 409A of the United States Internal Revenue Code of 1986, as amended (the “Code”). (b) Notwithstanding anything contained herein to the contrary, the Executive shall not be considered to have terminated employment with the Company for purposes of Section 6 and 7 of this Agreement unless he would be considered to have incurred a “separation from service” from the Company within the meaning of Treasury Regulation §1.409A-1(h)(1)(ii). (c) The severance payments set forth in Section 2(b) of this Agreement are intended to comply with the involuntary separation pay plan exception to Section 409A set forth in Treas. Reg. §1.409A-1(b)(9)(iii) to the extent that the payments due under Section 2(b) are in the aggregate less than the lesser of two times the lesser of (i) the sum of the Executive’s annualized compensation based upon the annual rate of pay for services provided to the Company for the taxable year of the Company preceding the taxable year in which the Termination Date occurs (adjusted for any increase during that year that was expected to continue indefinitely if Executive had not separated from service) and (ii) the applicable compensation limit under Section 401(a)(17) of the Code for the year in which the Termination Date occurs shall be in accordance with as an involuntary separation plan. (d) The payment set forth in Section 2(c) of this Agreement is intended to comply with the short-term deferral exception to Section 409A of the Code set forth in Treas. Reg. §1.409A-1(b)(4). (e) All references in this Agreement to Executive’s termination of employment shall mean Executive’s “separation from service” within the meaning of Section 409A of the Code and Treas. Reg. §1.409A-1(h). (f) Executive acknowledges and agrees that notwithstanding any provision of this Agreement, the Company and its affiliates are not providing Executive with any tax advice with respect to Section 409A of the Code or otherwise and are not making any guarantees or other assurances of any kind to Executive with respect to the tax consequences or treatment of any amounts paid or payable to Executive under this Agreement. (g) Notwithstanding anything to the contrary contained herein, if necessary to comply with the restriction in Section 409A(a)(2)(B) of the Internal Revenue Code of 1986, as amended (the “Code”) concerning payments to “specified Executives,” any payment on account of the Executive’s separation from service that would otherwise be due hereunder within six (6) months after such separation shall nonetheless be delayed until the first business day of the seventh month following the Executive’s date of termination and the first such payment shall include the cumulative amount of any payments that would have been paid prior to such date if not for such restriction, together with interest on such cumulative amount during the period of such restriction at a rate, per annum, equal to the applicable federal short-term rate (compounded monthly) in effect under Section 1274(d) of the Code on the date of termination. If the Executive dies during the six- month postponement period prior to the payment, the amount of the payment deferred on account

of Section 409A of the Code shall be paid to the personal representative of the Executive’s estate within 30 days after the date of the Executive’s death. For purposes of this Section 5, the Executive shall be a “specified Executive” for the 12-month period beginning on the first day of the fourth month following each “Identification Date” if he is a “key Executive” (as defined in Section 416(i) of the Code without regard to Section 416(i)(5) thereof) of the Company at any time during the 12-month period ending on the “Identification Date.” For purposes of the foregoing, the Identification Date shall be December 31. (h) To the extent that any provision in this Agreement is ambiguous as to its compliance with Section 409A, the provision shall be read in such a manner so that no payments due under this Agreement shall be subject to an “additional tax” as defined in Section 409A(a)(1)(B) of the Code. (i) In no event whatsoever shall the Company be liable for any additional tax, interest or penalty that may be imposed on Executive by Section 409A or damages for failing to comply with Section 409A. Section 6. Section 280G Limitation. If any payment(s) or benefit(s) Executive would receive pursuant to this Agreement and/or pursuant to any other agreement or arrangement would (a) constitute a “parachute payment” within the meaning of Section 280G of the Code, (b) but for this Section 5, be subject to the excise tax imposed by Section 4999 of the Code (the “Excise Tax”), and (c) if the net-after tax amount (taking into account all applicable taxes payable by Executive, including any Excise Tax) that Executive would receive with respect to such payments or benefits does not exceed the Reduced Amount, then such payment(s) or benefit(s) (collectively, “Payments”) shall be reduced to the Reduced Amount. The “Reduced Amount” shall be the largest portion of the Payments that can be paid or provided without causing any portion of the Payments being subject to the Excise Tax. If a reduction in payments or benefits constituting “parachute payments” is necessary so that the Payments equal the Reduced Amount, reduction shall occur in the following order: (i) first, any base salary severance payments; (ii) second, any other cash payments due under any other agreement between the Company and Executive; (iii) third, cancellation of the acceleration of vesting of any stock options; (iv) fourth, cancellation of the acceleration of vesting of any restricted stock and restricted stock units; and (v) lastly, other non-cash forms of benefits. Calculations of the foregoing will be performed at the expense of the Company by an accounting firm selected by the Company. The determinations of such accounting firm shall be final, binding and conclusive upon the Company and Executive. Section 7. Severance Plan. For the avoidance of doubt, Executive’s severance rights arise solely under this Agreement. Executive is not a “Participant” in Lisata Therapeutics, Inc. Severance Plan, effective June 10, 2025 (the “Severance Plan”) and is not entitled to any benefits under the Severance Plan. Section 8. Binding Agreement; No Assignment. This Agreement shall be binding upon, and shall inure to the benefit of, Executive and the Company and their respective permitted successors, assigns, heirs, beneficiaries and representatives. This Agreement is personal to Executive and may not be assigned by Executive. This Agreement may not be assigned by the Company except in connection with a sale of all or substantially all of its assets or a merger or

consolidation of the Company, and the acquiring Company or entity expressly assumes this Agreement. Any attempted assignment in violation of this Section 8 shall be null and void. Section 9. Governing Law; Consent to Jurisdiction. The validity, interpretation, performance, and enforcement of this Agreement shall be governed by the laws of the State of New Jersey. In addition, Executive and the Company irrevocably submit to the exclusive jurisdiction of the courts of the State of New Jersey and the United States District Court sitting in New Jersey for the purpose of any suit, action, proceeding or judgment relating to or arising out of this Agreement and the transactions contemplated hereby. Service of process in connection with any such suit, action or proceeding may be served on Executive or the Company anywhere in the world by the same methods as are specified for the giving of notices under this Agreement. Executive and the Company irrevocably consent to the jurisdiction of any such court in any such suit, action or proceeding and to the laying of venue in such court. In any such action or proceeding, the court shall have the authority to award reasonable costs, expenses, and attorneys’ fees to the party that substantially prevails. Section 10. Entire Agreement; Amendments. This Agreement embodies the entire agreement between Executive and the Company with respect to the subject matter hereof and may only be amended or otherwise modified by a writing executed by all of the parties hereto. This Agreement supersedes all prior agreements and understandings, written or verbal, between Executive and the Company regarding the terms and conditions of the subject matter hereof, including the Prior Agreements. Section 11. Counterparts. This Agreement may be executed in any number of counterparts, each of which when executed shall be deemed to be an original and all of which together shall be deemed to be one and the same instrument. Section 12. Severability. If any provision, section or paragraph, or specific term contained therein, of this Agreement or the application thereof is determined by a court to be illegal, invalid or unenforceable, that provision, section, paragraph or term shall not be a part of this Agreement, and the legality, validity and enforceability of remaining provisions, sections and paragraphs, and all other terms therein, of this Agreement shall not be affected thereby. [Signature Page Follows]

LISATA THERAPEUTICS, INC. By: /s/ David J. Mazzo Name: David J. Mazzo, Ph.D. President and Chief Executive Officer Agreed to and Accepted: /s/ Tariq Imam Date: June 10, 2025
Exhibit 31.1
CERTIFICATIONS UNDER SECTION 302
I, David J. Mazzo, PhD, certify that:
1. I have reviewed this Quarterly Report on Form 10-Q of Lisata Therapeutics, Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under my supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to me by others within those entities, particularly during the period in which this report is being prepared;
b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under my supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c) Evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report my conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d) Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant's other certifying officer and I have disclosed, based on my most recent evaluation of internal control over financial reporting to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
Date: August 7, 2025
/s/ David J. Mazzo, PhD
Name: David J. Mazzo, PhD
Title: President & Chief Executive Officer
(Principal Executive Officer)
Exhibit 31.2
CERTIFICATIONS UNDER SECTION 302
I, James Nisco, certify that:
1. I have reviewed this Quarterly Report on Form 10-Q of Lisata Therapeutics, Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under my supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to me by others within those entities, particularly during the period in which this report is being prepared;
b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under my supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c) Evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report my conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d) Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant's other certifying officer and I have disclosed, based on my most recent evaluation of internal control over financial reporting to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
Date: August 7, 2025
/s/ James Nisco
Name: James Nisco
Title: Senior Vice President, Finance and Treasury and Chief Accounting Officer
(Principal Financial Officer and Principal Accounting Officer)
Exhibit 32.1
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Quarterly Report on Form 10-Q of Lisata Therapeutics, Inc. (the “Company”) for the quarter ended June 30, 2025 filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, David J. Mazzo, PhD, President and Chief Executive Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, to my knowledge that:
1.The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934 as amended; and
2.The information contained in the Report fairly presents, in all material respects, the financial condition of the Company as of the dates presented and the results of operations of the Company for the periods presented.
Dated: August 7, 2025
| /s/ David J. Mazzo, PhD | |||||
| David J. Mazzo, PhD | |||||
| President & Chief Executive Officer (Principal Executive Officer) | |||||
The foregoing certification is being furnished solely pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (subsections (a) and (b) of Section 1350, Chapter 63 of Title 18, United States Code) and is not being filed as part of the Form 10-Q or as a separate disclosure document.
Exhibit 32.2
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
In connection with the Quarterly Report on Form 10-Q of Lisata Therapeutics, Inc. (the “Company”) for the quarter ended June 30, 2025 filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, James Nisco, Senior Vice President, Finance and Treasury and Chief Accounting Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, to my knowledge that:
1.The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934 as amended; and
2.The information contained in the Report fairly presents, in all material respects, the financial condition of the Company as of the dates presented and the results of operations of the Company for the periods presented.
Dated: August 7, 2025
| /s/ James Nisco | |||||
| James Nisco | |||||
| Senior Vice President, Finance and Treasury and Chief Accounting Officer (Principal Financial Officer and Principal Accounting Officer) | |||||
The foregoing certification is being furnished solely pursuant to Section 906 of the Sarbanes-Oxley Act of 2002 (subsections (a) and (b) of Section 1350, Chapter 63 of Title 18, United States Code) and is not being filed as part of the Form 10-Q or as a separate disclosure document.
